Yael Abraham, Ph.D. Independent Chief Scientific Consultant
Singlet Oxygen Low-Dose Technology (patented in the U.S., South Korea, China, India, Mexico, Europe, and other countries worldwide) ORCID: 0009-0001-8793-0096
Contact: yael.abraham.phd@gmail.com
How to cite this version: Abraham, Y. (2026). Low-Dose Singlet Oxygen as a Hormetic Agent: Mechanisms, Physiological Effects, and Therapeutic Potential. Zenodo.
This review characterizes the role of singlet oxygen (¹O₂) as a non-pharmacological, hormetic agent, capable of activating adaptive stress responses to support cellular resilience without inducing oxidative damage. Low-dose gas-phase singlet oxygen acts as a biophysical trigger at the plasma membrane, initiating a systemic adaptive relay. We detail the biochemical mechanisms of this response, focusing on the Nrf2-Keap1-SIRT6 axis, MAPK-mediated kinase rheostats, and the induction of autophagic flux (mitophagy, lipophagy). The review contrasts traditional irradiative singlet oxygen generation with a novel, non-irradiative catalytic technology (U.S. Patent No. 11,007,129 B2), highlighting its advantages in maintaining a stable, sub-toxic output suitable for continuous therapeutic application. Integrated clinical observations across respiratory, sleep, and metabolic contexts provide preliminary evidence for the “systemic recalibration” induced by this technology.
Singlet Oxygen, Hormesis, Nrf2, Autophagy, Redox Signaling, SIRT6, Mitophagy, U.S. Patent 11,007,129 B2, Adaptive Stress Response, Non-irradiative Catalysis.
Disclosure
The author serves as an independent consultant for the patent registration and scientific aspects of the technology described herein. This work was conducted to provide a theoretical framework for observed physiological outcomes and received no direct commercial funding.
Redox biology has revolutionized our understanding of how organisms manage internal and environmental challenges. Reactive oxygen species (ROS), once viewed exclusively as harmful metabolic byproducts, are now recognized as essential signaling molecules that modulate adaptive stress responses, immune regulation, and cellular repair. Among these ROS, singlet oxygen (¹O₂), an electronically excited form of molecular oxygen, plays a unique dual role as both a cytotoxic agent at high concentrations and a physiological regulator at controlled low levels.[i]
Singlet oxygen is naturally generated through enzymatic reactions, mitochondrial activity, and photoactivation processes in plants[ii] and animals[iii], influencing diverse physiological and pathological pathways. Its effects depend strongly on concentration and exposure duration: while excessive levels cause oxidative damage, sub-toxic doses can activate hormetic mechanisms that enhance resilience, reduce inflammation, and support regeneration.[iv]
Recent advances in delivery technologies, particularly non-irradiative gas-phase catalytic generation, offer new opportunities to administer low-dose singlet oxygen in a controlled, non-invasive manner, potentially extending its application from laboratory and photodynamic contexts to broader preventive and wellness domains. This approach circumvents the limitations of photosensitizer-based systems, such as photobleaching, optical alignment constraints, and environmental sensitivity, while enabling stable output for long-term use.
While the hormetic properties of other ROS are well established, the specific signaling and systemic effects of continuous low-dose singlet oxygen exposure in humans remain insufficiently studied. There is a lack of controlled clinical data defining optimal dosing, safety margins, and mechanistic biomarkers for long-term application. The present review integrates current mechanistic knowledge with observational clinical reports to outline plausible biological pathways, highlight Physiological potential, and identify priorities for future research.
Through this comprehensive lens, singlet oxygen is positioned as a distinctive agent in redox biology, bridging environmental sensing and systemic resilience, and warranting further exploration as a novel hormetic stimulus in human health.
This review explores the potential of low-dose singlet oxygen as a hormetic agent that activates adaptive stress responses without inducing oxidative damage. We examine the molecular pathways it influences, its role in organ and systemic regulation, and the supportive role in biological maintenance of these effects. Importantly, we highlight a novel, non-irradiative method for delivering singlet oxygen and review both mechanistic evidence and case observations supporting its use.
[i] (a) Klotz, L. O., Kröncke, K. D., & Sies, H. (2003). Singlet oxygen-induced signaling effects in mammalian cells. Photochemical & photobiological sciences, 2(2), 88-94. (b) Jones, D. P., & Sies, H. (2015). The redox code. Antioxidants & redox signaling, 23(9), 734-746.
[ii] Zheng, C., Chen, J. P., Wang, X. W., & Li, P. (2025). Reactive Oxygen Species in Plants: Metabolism, Signaling, and Oxidative Modifications. Antioxidants, 14(6), 617.
[iii] Murotomi, K., Umeno, A., Shichiri, M., Tanito, M., & Yoshida, Y. (2023). Significance of singlet oxygen molecule in pathologies. International Journal of Molecular Sciences, 24(3), 2739.
[iv] Calabrese, E. J., & Mattson, M. P. (2011). Hormesis provides a generalized quantitative estimate of biological plasticity. Journal of cell communication and signaling, 5(1), 25-38.
Oxygen constitutes approximately 20% of atmospheric air and is vital for aerobic life. In its most stable form, dioxygen (O₂) exists as a diradical in the ground triplet state (³Σg⁻), characterized by two unpaired electrons in separate degenerate orbitals (Figure 1a). This electronic configuration imposes spin restrictions on reactions with singlet-state molecules (which most biological molecules are) but allows reactivity with radicals.
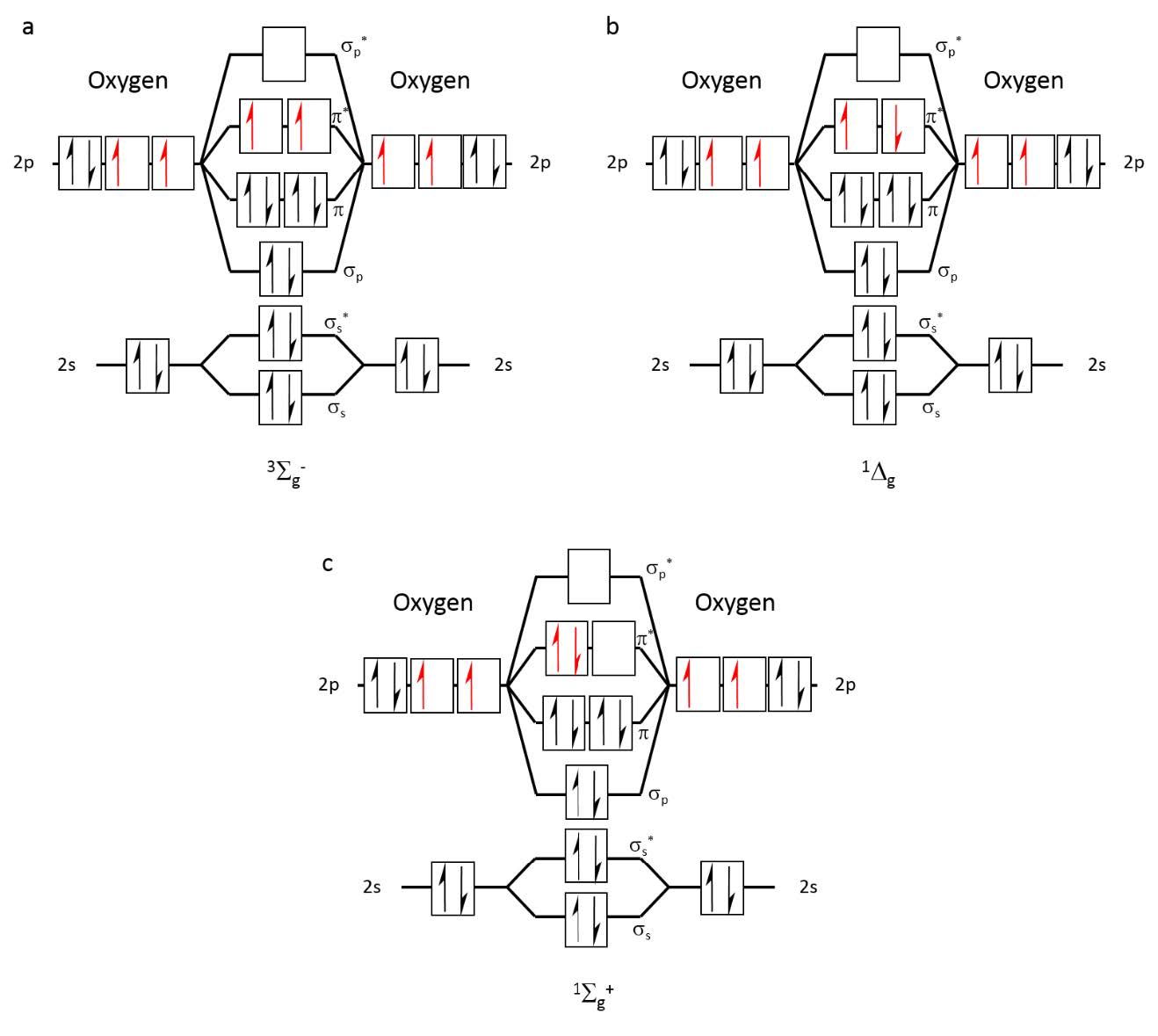
Figure 1 Occupation of molecular orbitals in oxygen at different energetic states: (a) triplet ground state, 3Sg–, (b) Most stable singlet state, 1Dg, (c) Highest energy, short- lived singlet state, 1Σg+.
Excitation of molecular oxygen leads to two singlet states: ¹Δg and ¹Σg⁺ (Figure 1b and Figure 1c, respectively). The higher-energy ¹Σg⁺ state is extremely reactive but short-lived, rapidly relaxing to the lower-energy ¹Δg state, commonly referred to as singlet oxygen (¹O₂). In the gas phase, isolated singlet oxygen (¹Δg) has a relatively long lifetime (~72 minutes),[1] but this decreases significantly with increased probability of collisions in liquids or under higher pressure/temperature. Under these conditions, the lifetime can reduce to mere seconds in the gas phase.[2] In solutions, the lifetime of singlet oxygen is even shorter, ranging from microseconds to nanoseconds, depending on the solvent properties.[3]
Singlet oxygen can be artificially produced through a range of methods, with most traditional techniques relying on liquid-phase systems. One of the most established is chemical generation,[iv] which involves reactions such as the decomposition of trioxidane in water or reaction of hydrogen peroxide with sodium hypochlorite (Figure 2). This method is effective in liquids but produces reactive byproducts and has short diffusion range. Another common method is photosensitization,[v] in which light-excitable compounds (photosensitizers) such as porphyrins transfer energy to ground-state triplet oxygen, producing singlet oxygen via a type II energy transfer mechanism (Figure 3). This light-driven process is the foundation of photodynamic therapy (PDT), a clinically validated modality used in the treatment of certain cancers,[vi] microbial infections,[vii] and dermatological disorders.[viii] Despite its efficacy, photosensitized singlet oxygen production requires localized light exposure, often limiting treatment to surface tissues or necessitating invasive delivery systems. Moreover, outcomes are influenced by complex pharmacokinetics, oxygen availability, and potential phototoxicity.[ix] Other methods include Plasma-induced excitation which generates mixed ROS, including singlet oxygen, without photosensitizers and laser/microwave excitation which is used primarily in specialized high-energy applications.
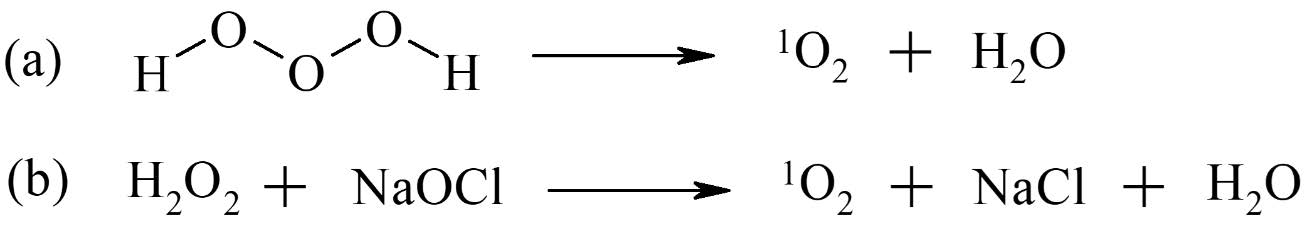
Figure 2 Examples for chemical reactions to produce singlet oxygen, 1O2: (a) decomposition of trioxidane in water; (b) reaction of hydrogen peroxide with sodium hypochlorite.
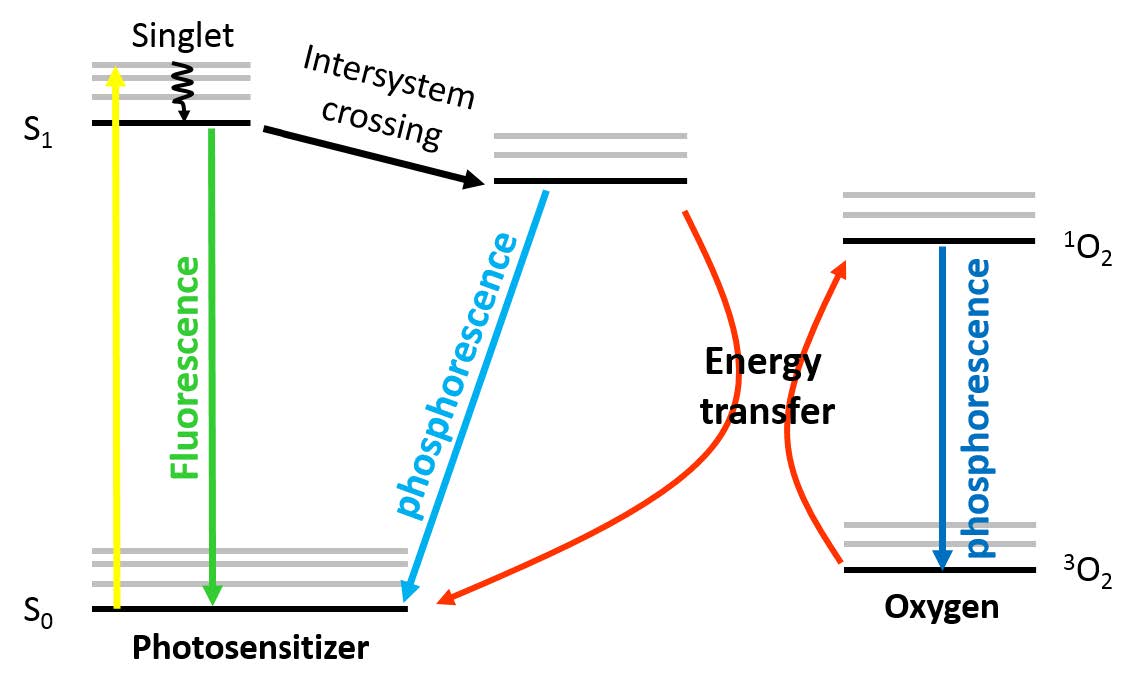
Figure 3 Energy diagram illustrating singlet oxygen (1O2) that is generated from ground state triplet oxygen (3O2) via energy transfer from the excited state of a photosensitizer to the oxygen molecule upon irradiation.
To expand singlet oxygen applications beyond solution-phase systems, alternative gas-phase generation strategies have been developed.
Gas-phase singlet oxygen has a longer intrinsic lifetime and greater diffusion capacity compared with singlet oxygen generated in liquid or solution-constrained environments.6 These properties enable non-invasive delivery across larger biological or environmental surfaces and remove the need for liquid media, photonic targeting, or pharmacokinetic considerations.
Historically, the most common method is photosensitization, in which immobilized dyes (e.g., porphyrins, Rose Bengal) absorb light and transfer energy to ground-state oxygen.[x] While effective in laboratory settings, these irradiative systems face significant challenges for continuous or low-dose clinical use. Organic sensitizers are prone to photobleaching, which is a gradual loss of catalytic efficiency due to light-induced degradation, which can lead to inconsistent dosing over time. Furthermore, irradiative systems are sensitive to humidity and temperature fluctuations and require precise optical alignment, limiting their scalability for unattended domestic or long-term therapeutic operation. [xi]
Other methods include plasma-based generation, which employs non-thermal plasma to excite molecular oxygen, without the need for light or sensitizers. However, plasma methods often generate a broad, high-energy mixture of reactive oxygen species (ROS), including ozone (O3) and nitrogen oxides (NOx), which can introduce collateral toxicity that overrides the desired hormetic effect.[xii] Similarly, heterogeneous photocatalysis using doped metal oxides (e.g., doped TiO₂, ZnO) generate singlet oxygen from atmospheric oxygen under visible light or UV, which may carry secondary irradiative risks. [xiii]
A novel, non-irradiative catalytic approach (as detailed in U.S. Patent No. 11,007,129 B2)[xiv] produces gas-phase singlet oxygen by passing ambient air through engineered metal-based substrates. This method utilizes specific metal alloys with controlled oxygen affinity to facilitate a “soft” electronic excitation.[xv] In this process, oxygen molecules are transiently adsorbed and excited to the singlet state before release, without fully oxidizing the metal surface or requiring an external light source.
The primary advantage of this patented catalytic principle is its energy purity and stability. By bypassing high-energy plasma and degradable photosensitizers, the system avoids the production of high-toxicity oxidants and maintains a constant, reproducible output. This technical precision allows for the delivery of a “sub-threshold” or low-dose signal that is inherently self-limiting. By controlling airflow parameters and catalytic residence time, the technology ensures the output remains within the hormetic zone- providing enough oxidative stimulus to trigger cellular recalibration (e.g., Nrf2 and autophagy pathways) without reaching the levels of oxidative stress that induce apoptosis or tissue damage.
Consequently, while traditional gas-phase systems are well established for industrial disinfection, these non-irradiative catalytic platforms offer a light-independent, low-maintenance alternative uniquely suited for therapeutic contexts. The physiological implications of this controlled, “pure” singlet oxygen signal are explored in the subsequent sections of this review.
[1] Wilkinson, F., Helman, W. P., & Ross, A. B. (1995). Rate constants for the decay and reactions of the lowest electronically excited singlet state of molecular oxygen in solution. An expanded and revised compilation. Journal of Physical and Chemical Reference Data, 24(2), 663-677.
[2] (a) Hasegawa, K., Yamada, K., Sasase, R., Miyazaki, R., Kikuchi, A., & Yagi, M. (2008). Direct measurements of absolute concentration and lifetime of singlet oxygen in the gas phase by electron paramagnetic resonance. Chemical Physics Letters, 457(4-6), 312-314. (b) Wang, K. K., Song, S., Jung, S. J., Hwang, J. W., Kim, M. G., Kim, J. H., … & Kim, Y. R. (2020). Lifetime and diffusion distance of singlet oxygen in air under everyday atmospheric conditions. Physical Chemistry Chemical Physics, 22(38), 21664-21671.
[3] Hurst, J. R., McDonald, J. D., & Schuster, G. B. (1982). Lifetime of singlet oxygen in solution directly determined by laser spectroscopy. Journal of the American Chemical Society, 104(7), 2065-2067.
[iv] (a) Noronha-Dutra, A. A., Epperlein, M. M., & Woolf, N. (1993). Reaction of nitric oxide with hydrogen peroxide to produce potentially cytotoxic singlet oxygen as a model for nitric oxide-mediated killing. FEBS letters, 321(1), 59-62. (b) Kanofsky, J. R. (1984). Singlet oxygen production by chloroperoxidase-hydrogen peroxide-halide systems. Journal of Biological Chemistry, 259(9), 5596-5600. (c) Foote, C. S., Wexler, S., Ando, W., & Higgins, R. (1968). Chemistry of singlet oxygen. IV. Oxygenations with hypochlorite-hydrogen peroxide. Journal of the American Chemical Society, 90(4), 975-981. (d) Stephenson, L. M., & McClure, D. E. (1973). Mechanisms in phosphite ozonide decomposition to phosphate esters and singlet oxygen. Journal of the American Chemical Society, 95(9), 3074-3076.
[v] Barry Halliwell; John MC. (1982) Free Radical in Biology and Medicine. Second Edition. Clarwndon Press. OxFord.
[vi] Makuch, S., Dróżdż, M., Makarec, A., Ziółkowski, P., & Woźniak, M. (2022). An Update on Photodynamic Therapy of Psoriasis—Current Strategies and Nanotechnology as a Future Perspective. International journal of molecular sciences, 23(17), 9845.
[vii] Jiang, J., Lv, X., Cheng, H., Yang, D., Xu, W., Hu, Y., … & Zeng, G. (2024). Type I photodynamic antimicrobial therapy: principles, progress, and future perspectives. Acta Biomaterialia, 177, 1-19.
[viii] Singh, S., & Awasthi, R. (2023). Breakthroughs and bottlenecks of psoriasis therapy: Emerging trends and advances in lipid based nano-drug delivery platforms for dermal and transdermal drug delivery. Journal of Drug Delivery Science and Technology, 104548.
[ix] DeRosa, M. C., & Crutchley, R. J. (2002). Photosensitized singlet oxygen and its applications. Coordination Chemistry Reviews, 233, 351-371.
[x] (a) Eisenberg, W. C., & DeSilva, M. (1990). Atmospheric gas phase generation of singlet oxygen by homogeneous photosensitization. Tetrahedron letters, 31(41), 5857-5860. (b) Eisenberg, W. C., & DeSilva, M. (1990). Atmospheric gas phase generation of singlet oxygen by homogeneous photosensitization. Tetrahedron letters, 31(41), 5857-5860. (c) Funken, K. H., Horneck, G., Milow, B., Schafer, M., Schmitz, C., Faust, D., … & Sattlegger, M. (2000). U.S. Patent No. 6,107,480. Washington, DC: U.S. Patent and Trademark Office. (d) Sunday, M. O., & Sakugawa, H. (2020). A simple, inexpensive method for gas-phase singlet oxygen generation from sensitizer-impregnated filters: Potential application to bacteria/virus inactivation and pollutant degradation. Science of the Total Environment, 746, 141186.
[xi] (1) Ihalagedara, H. B., Xu, Q., Greer, A., & Lyons, A. M. (2025). Singlet oxygen generation on a superhydrophobic surface: Effect of photosensitizer coating and incident wavelength on 1O2 yields. Photochemistry and Photobiology, 101(1), 167-179. (2) Arpa, E. M., & Corral, I. (2023). Unveiling Photodegradation and Photosensitization Mechanisms of Unconjugated Pterins. Chemistry–A European Journal, 29(29), e202300519.
[xii] (a) Gorbanev, Y., & Bogaerts, A. (2018). Chemical detection of short-lived species induced in aqueous media by atmospheric pressure plasma. In Atmospheric Pressure Plasma-from Diagnostics to Applications. IntechOpen. (b) Cabrellon, G., Tampieri, F., Rossa, A., Barbon, A., Marotta, E., & Paradisi, C. (2020). Application of fluorescence-based probes for the determination of superoxide in water treated with air non-thermal plasma. ACS sensors, 5(9), 2866-2875. (c) Aggelopoulos, C. A., Tataraki, D., & Rassias, G. (2018). Degradation of atrazine in soil by dielectric barrier discharge plasma–potential singlet oxygen mediation. Chemical Engineering Journal, 347, 682-694. (d) Jablonowski, H., Santos Sousa, J., Weltmann, K. D., Wende, K., & Reuter, S. (2018). Quantification of the ozone and singlet delta oxygen produced in gas and liquid phases by a non-thermal atmospheric plasma with relevance for medical treatment. Scientific reports, 8(1), 12195. (e) Lim, J., Park, S., Ryu, S., Park, S., & Kim, M. S. (2025). Different inactivation mechanisms of Staphylococcus aureus and Escherichia coli in water by reactive oxygen and nitrogen species generated from an argon plasma jet. Environmental Science & Technology, 59(6), 3276-3285.
[xiii] (a) Kuk, S. K., Ji, S. M., Kang, S., Yang, D. S., Kwon, H. J., Koo, M. S., … & Lee, H. C. (2023). Singlet-oxygen-driven photocatalytic degradation of gaseous formaldehyde and its mechanistic study. Applied Catalysis B: Environmental, 328, 122463. (b) Zollo, A., Livraghi, S., Giamello, E., Cioni, A., Dami, V., Lorenzi, G., … & Zaleska-Medynska, A. (2023). How to guide photocatalytic applications of titanium dioxide co-doped with nitrogen and carbon by modulating the production of reactive oxygen species. Journal of Environmental Chemical Engineering, 11(6), 111523. (c) Li, Q., Zhao, J., Shang, H., Ma, Z., Cao, H., Zhou, Y., … & Li, H. (2022). Singlet Oxygen and Mobile Hydroxyl Radicals Co-operating on Gas–solid Catalytic Reaction Interfaces for Deeply Oxidizing NO x. Environmental Science & Technology, 56(9), 5830-5839. (d) Ibhadon, A. O., & Fitzpatrick, P. (2013). Heterogeneous photocatalysis: recent advances and applications. Catalysts, 3(1), 189-218. (e) Shi, Y., Yang, Z., Shi, L., Li, H., Liu, X., Zhang, X., … & Zhang, L. (2022). Surface boronizing can weaken the excitonic effects of BiOBr nanosheets for efficient O2 activation and selective NO oxidation under visible light irradiation. Environmental Science & Technology, 56(20), 14478-14486. (f) Yaghmaei Sabegh, M. (2024). Advancements in Photochemistry and Flow Systems for Synthesis and Analysis (Doctoral dissertation, Université d’Ottawa| University of Ottawa).
[xiv] Badash, Z. (2021). U.S. Patent No. 11,007,129. Washington, DC: U.S. Patent and Trademark Office.
[xv] Carbogno, C., Groß, A., Meyer, J., & Reuter, K. (2013). O2 Adsorption Dynamics at Metal Surfaces: Non-Adiabatic Effects, Dissociation and Dissipation. In Dynamics of Gas-Surface Interactions (pp. 389-419). Springer Berlin Heidelberg.
Singlet oxygen, as mentioned before, is considered to be an ROS and is a natural product of various biological processes in both animals and plants. For instance, photosensitization can also occur in plants,[i] as chlorophyll, that is the green pigment essential for photosynthesis in leaves, can function as a natural photosensitizer, especially under high light intensity. At these conditions, the chlorophyll molecules facilitate the formation of singlet oxygen, which acts as a signaling molecule, as it triggers photoinhibition, a protective mechanism that prevents damage to the photosynthetic apparatus from excessive light exposure. [ii]
More commonly, singlet oxygen is mainly generated endogenously through light-independent pathways. In non-photosensitized tissues, enzymatic systems such as myeloperoxidase (MPO) in neutrophils and eosinophils can catalyze reactions involving hydrogen peroxide and chloride ions, leading to the formation of hypochlorous acid and subsequently singlet oxygen.[iii] In addition, singlet oxygen is generated within different cellular compartments, such as peroxisomes, the endoplasmic reticulum, the cytosol, and, most notably, mitochondria. [iv] Mitochondria, the organelles responsible for energy (ATP) production through aerobic respiration, use oxygen as an electron source. This process results in the formation of ROS, including hydrogen peroxide (H₂O₂), hydroxyl radicals (OH•), superoxide anion radicals (O₂•⁻), and singlet oxygen (¹O₂). Interestingly, it has been shown that singlet oxygen at controlled levels may enhance mitochondrial activity and energy metabolism. [v] These internal sources suggest that singlet oxygen is not confined to exogenous or photodynamic contexts but may act as an intrinsic modulator of redox signaling under physiological or inflammatory conditions.
The biological activity of singlet oxygen is defined by a delicate balance between concentration and exposure duration. The constant production of singlet oxygen in the body leads to a baseline concentration estimated to be around 10⁻¹³ M under neutral conditions, which is far below the threshold for cellular damage. In contrast, concentrations reaching approximately 10⁻⁸ M can damage cell membranes, while local concentrations of 10⁻⁵–10⁻⁴M are associated with irreversible oxidative damage and cell death.[vi]
Cellular and tissue damage occurs when ROS, including singlet oxygen, oxidize electron-rich sites, such as double bonds and thiol groups, in key biomolecules (Error! Reference source not found.). Critical cellular targets include pigments and antioxidants (e.g., chlorophyll, hemoglobin), proteins (e.g., enzymes, structural and transport proteins, receptors),[vii] lipids (e.g., membrane phospholipids), and nucleic acids.[viii] Damage to these macromolecules disrupts cellular integrity and function, contributing to pathological conditions.
The concept of oxidative stress arises when reactive species like singlet oxygen accumulate beyond the buffering capacity of cellular antioxidant systems. This threshold-based response underscores the necessity of maintaining a redox equilibrium and opens the door for hormetic interventions, where sub-threshold exposures may prime cellular defenses rather than cause harm.
Understanding these concentration-response relationships is essential for the safe therapeutic application of singlet oxygen and provides a framework for distinguishing between beneficial and deleterious exposure. Having outlined the toxicity thresholds of singlet oxygen, we now turn to the beneficial cellular processes activated within the safe, sub-toxic range, processes that exemplify the concept of redox hormesis.
Table 1 Chemical Activity of Singlet Oxygen With Various Biomolecules.21,25–27
Biomolecule Type | Key Reactive Groups | Reaction Type | Example Products | Biological Outcomes |
Lipids (PUFAs) | Allylic C=C double bonds | Ene reaction | Lipid hydroperoxides (e.g., 13-HPODE) | Membrane damage, lipid peroxidation chain reactions, formation of signaling aldehydes (e.g., 4-HNE), inflammation |
Proteins | Trp, His, Met, Cys | Oxidation of side chains | Methionine sulfoxide, N-formylkynurenine, 2-oxo-histidine | Loss of enzyme activity, altered protein folding, proteasomal degradation, redox signaling |
Nucleic Acids | Guanine (mainly) | Base oxidation | 8-oxo-7,8-dihydroguanine (8-oxoG) | Mutagenesis, transcriptional errors, impaired DNA replication, contributes to age-related diseases and cancer |
Cholesterol | Allylic hydrogens, Δ5 double bond | Addition/Oxidation | 7-hydroperoxycholesterol, 25-hydroxycholesterol | Altered membrane structure, LXR pathway modulation, potential atherogenesis, implicated in inflammatory diseases |
Pigments and antioxidants (e.g., carotenoids, tocopherols) | Conjugated π-systems | Physical/chemical quenching | Inert or less-reactive byproducts | Impair function and protection from oxidative stress |
[i] Devasagayam, T., & Kamat, J. P. (2002). Biological significance of singlet oxygen.
[ii] Dmitrieva, V. A., Tyutereva, E. V., & Voitsekhovskaja, O. V. (2020). Singlet oxygen in plants: Generation, detection, and signaling roles. International journal of molecular sciences, 21(9), 3237.
[iii] Kiryu, C., Makiuchi, M., Miyazaki, J., Fujinaga, T., & Kakinuma, K. (1999). Physiological production of singlet molecular oxygen in the myeloperoxidase-H2O2-chloride system. FEBS letters, 443(2), 154-158.
[iv] Sun, Y., Lu, Y., Saredy, J., Wang, X., Drummer IV, C., Shao, Y., … & Yang, X. (2020). ROS systems are a new integrated network for sensing homeostasis and alarming stresses in organelle metabolic processes. Redox biology, 37, 101696.
[v] Sokolovski, S. G., Rafailov, E. U., Abramov, A. Y., & Angelova, P. R. (2021). Singlet oxygen stimulates mitochondrial bioenergetics in brain cells. Free Radical Biology and Medicine, 163, 306-313.
[vi] Szechyńska-Hebda, M., Ghalami, R. Z., Kamran, M., Van Breusegem, F., & Karpiński, S. (2022). To Be or Not to Be? Are Reactive Oxygen Species, Antioxidants, and Stress Signalling Universal Determinants of Life or Death? Cells, 11(24), 4105.
[vii] Davies, M. J. (2004). Reactive species formed on proteins exposed to singlet oxygen. Photochemical & Photobiological Sciences, 3, 17-25.
[viii] (a) Di Mascio, P., Martinez, G. R., Miyamoto, S., Ronsein, G. E., Medeiros, M. H., & Cadet, J. (2019). Singlet molecular oxygen reactions with nucleic acids, lipids, and proteins. Chemical reviews, 119(3), 2043-2086. (b) Juan, C. A., Pérez de la Lastra, J. M., Plou, F. J., & Pérez-Lebeña, E. (2021). The chemistry of reactive oxygen species (ROS) revisited: outlining their role in biological macromolecules (DNA, lipids and proteins) and induced pathologies. International Journal of Molecular Sciences, 22(9), 4642.
Every biological system, from single cells to complex multicellular organisms, relies on maintaining specific internal conditions to function optimally. These conditions include stable temperature, pH, and precise concentrations of minerals and biomolecules, collectively referred to as “homeostasis.” However, as both external and internal environments are constantly changing, organisms have evolved specialized mechanisms to sustain homeostasis.
When an organism’s intrinsic mechanisms are unable to maintain stable internal conditions, the system enters a state of “stress.” This failure disrupts the optimal functioning of biological systems and, over time, leads to structural and functional damage. Various biomolecules and biostructures suffer chemical and physical damage, which might lead to various diseases and pathological syndromes.
Given that most organisms inhabit dynamic and often volatile environments, the induction of stress is inevitable. To survive and thrive, organisms must develop adaptive mechanisms to mitigate stress and restore homeostasis.
Oxidative stress is one of the most prevalent forms of stress experienced by organisms, particularly those relying on aerobic respiration. It arises when the ROS levels exceed the capacity of baseline antioxidant defenses to neutralize them. Elevated ROS levels pose a significant risk, as they can oxidize critical biomolecules altering their chemical and structural properties. Such changes often render these molecules ineffective, and in some cases, even harmful. As a result, at high levels, oxidative species are harmful to the body and may result in diseases.[i]
The potential for cellular and tissue damage underscores the importance of preventing ROS from reaching harmful levels. It has been found that at low concentrations, ROS essentially act as signalling molecules, as they activate cellular stress-response pathways1 that include the upregulation of antioxidant defences and mechanisms to repair existing damage. In this way, ROS play a crucial role in supporting cellular health and adaptability. By acting as signalling molecules, they not only alert the system to potential threats but also help initiate processes that restore balance and promote resilience in the face of environmental and physiological challenges.
Singlet oxygen is increasingly recognized not merely as a byproduct of oxidative stress but as a key signaling molecule in cellular regulation. Its role has been demonstrated across diverse biological systems, including plants[ii],[iii], bacteria[iv] and mammals20. One of its primary sites of action is the plasma membrane, where it chemically modifies specific lipids and proteins that function as primary oxidative targets, effectively acting as that trigger or inhibit downstream processes (.24,[v]
Among the most reactive are polyunsaturated fatty acids (PUFAs), whose oxidation produces stable secondary messengers like 4-hydroxynonenal (4-HNE). These messengers activate redox-sensitive transcription factors such as Nrf2 and NF-κB,[vi] initiating an antioxidant response that extends beyond the individual cell via paracrine signaling.
Furthermore, cholesterol, a major component of lipid rafts, is readily oxidized by singlet oxygen to form cholesterol hydroperoxides and oxysterols, such as 7-ketocholesterol and 25-hydroxycholesterol. These modulate lipid raft dynamics and membrane signaling.[vii] The oxidized derivatives influence membrane fluidity and can engage Liver X Receptors (LXRs) and Toll-like receptors (TLRs), amplifying metabolic and immunological responses.
Crucially, singlet oxygen oxidizes cysteine and methionine residues on membrane proteins, modulating ion channels and receptor conformations[viii] Together, these modifications convert the plasma membrane into a dynamic redox-sensitive signaling platform.
These modifications (summarized in Table 2) convert the plasma membrane into a dynamic, redox-sensitive signaling platform. The transformation of these localized membrane-centered redox events into a coordinated organism-level response necessitates a multi-tiered signal propagation system. This transition from a surface biophysical trigger to systemic adaptation is governed by three interconnected layers of communication. First, the intensity of the initial oxidative cue is decoded by a cytoplasmic kinase rheostat, primarily the ASK1-MAPK axis, which dictates the threshold between survival-oriented autophagy and apoptosis (see Section 5.3.1). Second, this signal is amplified and transported to distant tissues via extracellular vesicles and paracrine signaling, enabling a localized respiratory stimulus to influence systemic energy and sleep patterns. Finally, rapid physiological shifts are facilitated by neuroimmune relay systems that bypass slower transcriptional pathways to provide immediate homeostatic recalibration. The specific mechanics of these long-range propagation vehicles, and their role in bridging local sensing with systemic physiology, are detailed in Section 8.
Table 2 Molecular Targets in The Plasma Membrane That Act as ‘Biophysical Sensors’ For Singlet Oxygen
Molecular Target | Primary Modification | Systemic Significance |
PUFAs / Lipids | 4-HNE Production | Initiation of Paracrine/Vesicular Signaling |
Cholesterol | Oxysterol Formation | Modulation of Lipid Rafts and TLR activity |
Membrane Proteins | Thiol/Methionine Oxidation | Activation of Ion Channels and Neural Reflexes |
[i] (a) Forman, H. J., & Zhang, H. (2021). Targeting oxidative stress in disease: promise and limitations of antioxidant therapy. Nature Reviews Drug Discovery, 20(9), 689-709. (b) Brieger, K., Schiavone, S., Miller Jr, F. J., & Krause, K. H. (2012). Reactive oxygen species: from health to disease. Swiss medical weekly, 142, w13659. (c) Rahman, T., Hosen, I., Islam, M. T., & Shekhar, H. U. (2012). Oxidative stress and human health. Advances in Bioscience and Biotechnology, 3(07), 997.
[ii] Dmitrieva, V. A., Tyutereva, E. V., & Voitsekhovskaja, O. V. (2020). Singlet Oxygen in Plants: Generation, Detection, and Signaling Roles. International journal of molecular sciences, 21(9), 3237.
[iii] Saed-Moucheshi, A., Sohrabi, F., & Shirkhani, A. (2023). A review on reactive oxygen species (ROS): production, function, and their influence on plants. Crop Biotechnology, 13(44), 53-70.
[iv] Ziegelhoffer, E. C., & Donohue, T. J. (2009). Bacterial responses to photo-oxidative stress. Nature Reviews Microbiology, 7(12), 856-863.
[v] Maharjan, P. S., & Bhattarai, H. K. (2022). Singlet oxygen, photodynamic therapy, and mechanisms of cancer cell death. Journal of oncology, 2022(1), 7211485.
[vi] Poli, G., Leonarduzzi, G., Biasi, F., & Chiarpotto, E. (2004). Oxidative stress and cell signalling. Current medicinal chemistry, 11(9), 1163-1182.
[vii] (a) Iuliano, L. (2011). Pathways of cholesterol oxidation via non-enzymatic mechanisms. Chemistry and physics of lipids, 164(6), 457-468. (b) Kulig, W., Olżyńska, A., Jurkiewicz, P., Kantola, A. M., Komulainen, S., Manna, M., … & Jungwirth, P. (2015). Cholesterol under oxidative stress—How lipid membranes sense oxidation as cholesterol is being replaced by oxysterols. Free radical biology and medicine, 84, 30-41.
[viii] (a) Lorenzen, I., Eble, J. A., & Hanschmann, E. M. (2021). Thiol switches in membrane proteins-Extracellular redox regulation in cell biology. Biological chemistry, 402(3), 253-269. (b) Bigelow, D. J., & Squier, T. C. (2011). Thioredoxin-dependent redox regulation of cellular signaling and stress response through reversible oxidation of methionines. Molecular Biosystems, 7(7), 2101-2109.
Singlet oxygen exerts concentration-dependent effects on biological systems. At elevated levels, it contributes to oxidative damage and cell death. However, at sub-toxic concentrations, singlet oxygen triggers a hormetic response, activating intrinsic cellular defense mechanisms by upregulation of antioxidant enzymes, redox-sensitive signaling, immune modulation, and autophagic processes. This hormetic response promotes resilience, repair, and redox homeostasis. [i]
This chapter outlines the molecular pathways and systems influenced by low-dose singlet oxygen, providing a mechanistic foundation for its Physiological potential.
One of the primary oxidative stress sensors activated by singlet oxygen is Nrf2 (Nuclear factor erythroid 2–related factor 2),[ii] which is a transcription factor that regulates the antioxidant defense system through the Nrf2-Keap1 pathway. A homodimer of Keap1 (Kelch-like ECH-associated protein 1) inhibits Nrf2 transcriptional activity by binding to its evolutionarily conserved N-terminal regulatory domain, thereby targeting Nrf2 for ubiquitin-proteasome-dependent degradation.[iii] Exposure to ROS or lipid-derived secondary messengers (such as 4-HNE), generated via the oxidation of primary membrane targets (as described in section 4.3), oxidize specific cysteine residues on Keap1. This triggers a conformational change in the Keap1 homodimer, disrupting its inhibitory ‘clamp’ on Nrf2 and preventing further ubiquitination and degradation. Freed Nrf2 translocates to the nucleus, where it binds to antioxidant response elements (AREs) in the promoter regions of detoxification and cytoprotective genes, promoting their transcription.
SIRT6, a NAD⁺-dependent histone deacetylase, supports the antioxidant response by enhancing Nrf2 stability and facilitating access to the regulatory regions of antioxidant genes. Upon oxidative stress, SIRT6 expression increases and contributes to Nrf2 activation by reducing its degradation, promoting its accumulation in the nucleus,[iv] and enabling robust transcription of antioxidant targets. SIRT6 also modulates chromatin structure, promoting an open configuration at specific Nrf2-responsive loci, which enhances recruitment of transcriptional machinery and efficient expression of antioxidant genes to counteract oxidative damage.[v]
Major downstream targets include:
Nrf2 upregulates the catalytic (GCLC) and modifier (GCLM) subunits of glutamate-cysteine ligase, the rate-limiting enzyme in glutathione (GSH) biosynthesis.[vi],[vii] GSH is a central redox buffer that neutralizes hydrogen peroxide, lipid peroxides,[viii] and electrophilic agents via enzymatic and non-enzymatic reactions.[ix] Critically, it maintains cellular proteins in their reduced thiol form, preventing aberrant disulfide cross-linking that can impair enzymatic activity and cellular signaling.[x] By preserving this thiol-disulfide balance, GSH supports proper protein folding, redox-sensitive signal regulation, and cytoskeletal organization.[xi]
Importantly, Nrf2-dependent enhancement of GSH synthesis also contributes to tissue repair mechanisms. Elevated GSH levels facilitate fibroblast migration, reduce oxidative delay in re-epithelialization, and accelerate wound closure under inflammatory conditions.[xii] Thus, GSH not only buffers oxidative insults but orchestrates a redox environment conducive to regeneration.
Nrf2 activation leads to the upregulation of multiple Phase II detoxifying enzymes, including NAD(P)H quinone oxidoreductase 1 (NQO1), heme oxygenase-1 (HO-1), and peroxiredoxins (PRDXs). These enzymes mitigate oxidative and electrophilic stress not by directly scavenging ROS, but by converting reactive intermediates into less harmful, excretable forms.
Together, these enzymes form a highly inducible frontline defense that complements glutathione systems and enhances the cell’s capacity to detoxify harmful byproducts of oxidative stress and inflammation.
Nrf2 supports redox homeostasis not only through antioxidant enzymes but also regulating enzymes that maintain thiol integrity and modulate lipid signaling. A core component of this system includes thioredoxin reductase 1 (TXNRD1) and superoxide dismutases (SOD1–3). SODs catalyse the dismutation of superoxide anions into hydrogen peroxide,[xvi] a less reactive species that is further detoxified by catalase[xvii] or glutathione peroxidase. TXNRD1 maintains thioredoxin in its reduced form, enabling it to reduce disulfide bonds in proteins, thus repairing them, and ensuring redox signal fidelity.[xviii]
Additionally, Nrf2 influences the metabolism of oxidized lipids through enzymes like ALOX15 (Arachidonate 15-lipoxygenase), which converts polyunsaturated fatty acids into mediators such as 15-HETE and lipoxins. These bioactive lipids help resolve inflammation, promote macrophage polarization toward anti-inflammatory phenotypes, and regulate redox-sensitive immune responses. [xix]
To complement its antioxidant functions, Nrf2 also supports proteasome activity under stress conditions by promoting the transcription of specific proteasome subunits. This enhances both 20S and 26S proteasomal capacity, which is especially important when misfolded or oxidized proteins accumulate.[xx] In addition, Nrf2-driven adaptation enhances the efficiency of ATP-independent degradation by the 20S core proteasome — a critical pathway in cells where autophagic function or ATP supply is limited.[xxi]
Such Nrf2–proteasome interplay is particularly relevant in non-dividing or long-lived cells, including muscle fibers and neurons, where sustained proteotoxic stress can disrupt cellular function.
In parallel, oxidative stress and thiol imbalance can trigger protein unfolding and aggregation. Nrf2 indirectly supports proteostasis by sustaining redox conditions that favor proper protein folding and by coordinating with heat shock proteins (HSPs), which refold or triage misfolded proteins. Heat shock protein 70 (HSP70)[xxii] assists in protein folding and prevents aggregation under stress, activating transcription factor 4 (ATF4)[xxiii] that coordinates the integrated stress response, regulating redox balance and cell survival. This redox–chaperone axis is particularly important in long-lived cells, such as neurons, where oxidative injury can destabilize protein structure and impair cellular function.[xxiv]
Through these pathways, Nrf2-linked redox systems contribute broadly to cellular stability in the face of oxidative stress. In redox-sensitive tissues like the nervous[xxv] and cardiovascular[xxvi] systems, these mechanisms support resilience by modulating stress signaling, protecting structural proteins, and promoting inflammatory resolution,[xxvii] without relying on direct ROS scavenging alone.
Nrf2 contributes to mitochondrial health and metabolic adaptation under oxidative stress by regulating transcriptional programs that coordinate mitochondrial biogenesis, quality control, and antioxidant defense. Key among these is PGC-1α (Peroxisome proliferator-activated receptor gamma coactivator 1-alpha), a central coactivator that stimulates the expression of nuclear respiratory factors (NRF1, NRF2/GABPA)[xxviii] and mitochondrial transcription factor A (TFAM), together promoting mitochondrial DNA replication and respiratory enzyme synthesis.[xxix]
Mitochondrial adaptation to low-dose singlet oxygen is not a result of direct molecular interaction, but rather a coordinated response to the membrane-initiated redox relay described in section 4.3. The secondary messengers generated at the plasma interface (e.g., 4-HNE) diffuse through the cytosol to interact with mitochondrial-resident proteins, such as DJ-1 (PARK7).
Acting as a redox sentinel, DJ-1 stabilizes the Nrf2 protein by further inhibiting its Keap1-mediated degradation, specifically shielding the mitochondria from potential oxidative injury.[xxx] This ‘mitohormetic’ signaling triggers a transcriptional program led by PGC-1α, which coordinates with SIRT6 and Nrf2 to promote mitochondrial biogenesis and enhance the expression of mitochondrial-specific antioxidants like SOD2 and GPX4, which are critical in detoxifying mitochondrial ROS and preventing lipid peroxidation.[xxxi]
In addition to the Nrf2–DJ-1 axis, the sirtuin family member SIRT6 serves as a critical co-regulator of mitochondrial homeostasis and metabolic resilience under oxidative stress.[xxxii] As mentioned above, SIRT6 functions as a “redox-sensor” that can directly interact with and stabilize Nrf2, thereby amplifying the expression of the aforementioned ARE-dependent genes, such as HO-1 and NQO1.40 Beyond its nuclear roles, SIRT6 is essential for maintaining mitochondrial quality control by preventing mitochondrial fragmentation and supporting efficient ATP production, which is a process that aligns with the activity of PGC-1α and AMPK in responding to transient hormetic signals.67,[xxxiii] By coordinating nuclear antioxidant defenses with mitochondrial bioenergetics, SIRT6 ensures that low-dose singlet oxygen exposure results in metabolic adaptation rather than energetic failure, reinforcing the systemic resilience characteristic of redox hormesis.[xxxiv]
By aligning nuclear antioxidant defenses with mitochondrial bioenergetics, this relay ensures that the cell increases its ATP production efficiency and respiratory capacity. Consequently, the transient biophysical trigger at the membrane is translated into long-term metabolic resilience, preventing mitochondrial fragmentation and ensuring energetic homeostasis during periods of physiological stress.
In addition to regulating antioxidant gene expression, Nrf2 plays a pivotal role in modulating inflammation. Upon activation, Nrf2 suppresses the expression of pro-inflammatory cytokines such as IL-6, TNF-α, and IL-1β by interfering with NF-κB signaling and by inducing anti-inflammatory mediators like HO-1 and ferritin.
The anti-inflammatory efficacy of Nrf2 is significantly augmented by its crosstalk with SIRT6, that serves as a potent repressor of pro-inflammatory gene expression.[xxxvi] While Nrf2 suppresses the transcription of cytokines, SIRT6 acts directly at the chromatin level by deacetylating Histone H3 at the promoters of NF-κB target genes. This ‘epigenetic silencing’ effectively dampens the inflammatory response even in the presence of persistent stress signals. Under hormetic conditions, low-dose singlet oxygen may enhance the stability of both Nrf2 and SIRT6, creating a synergistic anti-inflammatory axis that prevents the transition from acute, protective signaling to chronic, systemic inflammation.[xxxvii] This coordinated regulation is particularly relevant in age-related ‘inflammaging’, where the concurrent decline of Nrf2 and SIRT6 otherwise leaves tissues vulnerable to cytokine-mediated damage.
This modulation reduces systemic inflammation, limits immune overactivation, and contributes to tissue protection in conditions such as:
Thus, as summarized in Table 3, Nrf2 serves as a dual-function regulator, both antioxidant and anti-inflammatory, essential to the hormetic adaptation elicited by singlet oxygen.
Table 3 Integrated Defense Systems Regulated by the Nrf2-ARE Axis
Functional Category | Primary Enzymes/Factors | Physiological Outcome |
Glutathione System | GCLC, GCLM, GSH | Thiol-disulfide balance; accelerated wound closure and re-epithelialization. |
Phase II Detox | NQO1, HO-1, PRDXs | Neutralization of quinones; degradation of pro-oxidant heme; H₂O₂ buffering. |
Proteostasis | 20S/26S Proteasomes, HSP70 | Clearing of oxidized/misfolded proteins; prevention of protein aggregation. |
Mitochondria | PGC-1α, TFAM, DJ-1 | Mitochondrial biogenesis; protection of mtDNA; metabolic adaptation. |
Anti-Inflammatory | IL-6/TNF-α inhibition | Suppression of cytokine storms; resolution of “inflammaging.” |
Beyond the Nrf2-Keap1 pathway, the membrane-initiated relay activates several additional signaling pathways involved in stress response, inflammation regulation, cell death and repair mechanisms. These pathways ensure that cells maintain homeostasis, limit damage, and initiate adaptive processes during oxidative stress.
For example, the p53 tumor suppressor protein,[xlii] a key regulator of DNA repair and apoptosis, is activated in response to oxidative stress, helping cells manage or eliminate damage. Similarly, the MAPK family consists of several key protein kinases, including ERK1/2 (extracellular signal-regulated kinase), JNK (c-Jun N-terminal kinase), and p38 MAPK, which are activated through upstream stress sensors, including oxidative modifications of receptor proteins, mitochondrial ROS signaling, and changes in redox balance (Table 4).[xliii]
Through this multi-layered signaling network, the cell converts the initial biophysical trigger at the membrane into a sophisticated ‘decision-making’ process that favors repair, survival, and structural rejuvenation.
Table 4 MAPK Functional Specialization
Kinase Pathway | Primary Trigger | Major Biological Outcome |
ERK 1/2 | Membrane Receptor Oxidation | Tissue Repair & Proliferation (Wound healing/Skin smoothing) |
JNK / p38 | Redox Imbalance / Lipid Mediators | Stress Adaptation & Homeostatic Calibration |
p53 Axis | Redox-Sensitive Kinases | Genomic Stability & DNA Repair coordination |
The “Relay” logic holds that the cell does not wait for internal damage to reach a critical mass; instead, the biophysical trigger at the membrane initiates a preemptive “quality control” protocol, with the capacity to promote either cell survival or cell death. This duality is mediated through its ability to induce autophagy or apoptosis, two distinct yet interconnected processes that are often activated sequentially in response to stress. Autophagy, a highly conserved mechanism present in all eukaryotes, from unicellular organisms to mammals,[xlvi] is responsible for selectively degrading oxidized and damaged intracellular components, such as proteins and organelles (particularly mitochondria), via the lysosomal pathway. This process is essential for preserving cellular integrity and function during mild stress.[xlvii]
The outcome of oxidative stress is not binary but graded, depending on its intensity, duration, and localization.33,[xlviii],[xlix] At low to moderate levels, oxidative stress initiates adaptive responses, including Nrf2 activation and the induction of autophagy, enabling cells to restore homeostasis. In contrast, sustained or intense oxidative insults can overwhelm these defenses, tipping the balance toward apoptosis or necrosis (Figure 4).
As established in earlier sections, singlet oxygen acts as a metabolic rheostat. At low hormetic doses, secondary messengers (like 4-HNE) and redox-sensitive kinases (ASK1) favor the induction of Autophagy, a survival-oriented process that clears the “biological clutter” before it can trigger Apoptosis (cell death).[l] In addition, A key non-transcriptional effect of singlet oxygen is the oxidative modification of the Bcl-2 protein. This disruption releases Beclin-1, the “master switch” for autophagy initiation, allowing for rapid cellular response that bypasses the need for slower nuclear transcription. 82,83,[li]
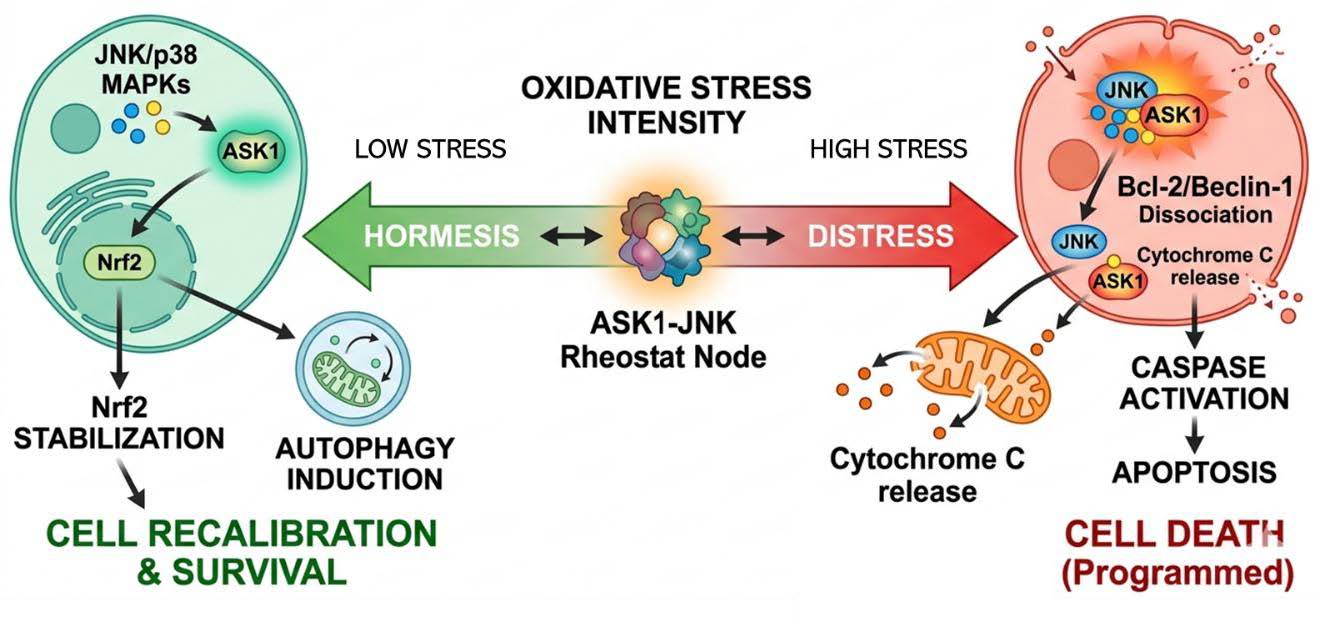
Figure 4 Schematic representation of the cellular decision-making process in response to varying intensities of singlet oxygen-induced stress. (Left) The Hormetic Zone: Under low-intensity oxidative conditions, the membrane-initiated relay activates a transient and moderate signaling surge through ASK1 and JNK/p38 MAPKs. This level of activation favors the stabilization of Nrf2 and the induction of Autophagy, specifically Mitophagy and CMA, to selectively sequester and degrade oxidatively damaged organelles and proteins. This ‘quality control’ phase promotes cellular recalibration and homeostatic recovery. (Center) The Rheostat Switch: The ASK1-JNK axis functions as a molecular rheostat, where the duration and amplitude of the oxidative signal determine the functional outcome. (Right) The Distress Zone: Under conditions of high-intensity or chronic oxidative stress, the sustained over-activation of these kinases leads to the phosphorylation-dependent dissociation of the Bcl-2–Beclin-1 complex and the subsequent release of pro-apoptotic factors, such as Cytochrome c. This shifts the cellular fate from survival-oriented cleanup to Caspase-dependent Apoptosis, ensuring the programmed elimination of irreversibly damaged cells. Together, these pathways illustrate the graded, non-binary nature of the stress response, where controlled oxidation acts as a primary messenger for survival.
Furthermore, singlet oxygen can influence the hypoxia-inducible factor 1 (HIF-1) pathway, which plays a pivotal role in cellular responses to low oxygen levels.[lii] While the HIF-1 pathway is primarily known for regulating genes that adapt to hypoxic conditions, it also intersects with autophagy mechanisms.
These immediate oxidative modifications allow singlet oxygen to act as a first messenger in stress adaptation, influencing the balance between repair and cell death independently of transcription. This reinforces the concept of oxidative stress as a rheostat, where controlled oxidation promotes survival pathways, while excess shifts the balance toward irreversible damage.
The primary forms of autophagy implicated are macroautophagy, mitophagy, and chaperone-mediated autophagy (CMA), each targeting distinct cellular liabilities within the cell.
Macroautophagy (often just referred to as autophagy) is responsible for bulk degradation of damaged or oxidized cytoplasmic material. Upon low-level oxidative stress, singlet oxygen can trigger macroautophagy by oxidizing redox-sensitive regulators, enabling the sequestration of dysfunctional proteins and organelles into autophagosomes. Although traditionally viewed as non-selective, macroautophagy often targets specific substrates based on oxidative modification patterns.82,[liii]
Mitophagy, a specialized subset of macroautophagy, mitophagy, specifically eliminates damaged or dysfunctional mitochondria. Mitochondria are central to cellular energy production. However, as organisms age, mitochondrial DNA (mtDNA) accumulates mutations, leading to decreased efficiency in the electron transport chain and increased production of ROS. The increased oxidative stress may further damage cellular components, thus accelerating aging and age-related diseases,[liv] including neurodegenerative disorders, [lv] cardiovascular diseases, [lvi] and metabolic syndromes[lvii]. This underscores the importance of inducing mitophagy to remove these compromised organelles and prevent further ROS production. Two mitophagy pathways have been identified. In one pathway, JNK influences mitophagy through the modulation of PINK1/Parkin Pathway, as JNK activation can promote the stabilization of PINK1 on the outer membrane of depolarized mitochondria, facilitating the recruitment of Parkin, an E3 ubiquitin ligase. Parkin ubiquitinates mitochondrial proteins, marking the mitochondria for autophagic degradation.[lviii]
Independently of the PINK/Parkin pathway, mitophagy can also occur through the phosphorylation of FUNDC1.[lix] JNK phosphorylates FUNDC1, a mitochondrial receptor, which enhances its binding affinity for LC3 or other autophagy proteins, facilitating the engulfment of damaged mitochondria by autophagosomes.
By utilizing both the PINK1/Parkin (ubiquitin-dependent) and FUNDC1 (receptor-dependent) pathways, the cell ensures that ‘leaky’ mitochondria, those with mutated mtDNA or inefficient electron transport, are selectively removed. This prevents the energetic failure typically seen in aging and metabolic syndromes, effectively ‘rejuvenating’ the cellular power supply.
Chaperone-Mediated Autophagy (CMA)[lx] is a form of autophagy that selectively degrades soluble cytosolic proteins containing the KFERQ-like pentapeptide motif. Under oxidative stress, oxidized proteins are recognized by chaperones and translocated directly into lysosomes for degradation. This process helps in the removal of oxidatively damaged proteins, thereby maintaining protein quality control.[lxi] JNK signaling has been implicated in modulating this pathway as it may influence the expression of chaperone proteins under mild oxidative conditions, thereby enhancing the selective degradation of oxidized cytosolic proteins. While the direct involvement of JNK in CMA regulation is less clear, oxidative stress may upregulate the expression of LAMP-2A,[lxii] the lysosomal receptor essential for CMA substrate translocation. Increased LAMP-2A levels enhance the cell’s capacity to degrade oxidized proteins via CMA.[lxiii] In addition, stress conditions can affect the activity of Hsc70,Error! Bookmark not defined. the cytosolic chaperone that recognizes and delivers substrates to LAMP-2A. Modulation of Hsc70 activity influences the efficiency of CMA.
Table 5 Specialized Autophagic Pathways Triggered by Oxidative Stress
Autophagy Subtype | Primary Target | Key Mediator/Relay |
Macroautophagy | Bulk oxidized proteins & lipids | Beclin-1 / ASK1 |
Mitophagy | Dysfunctional Mitochondria | JNK à PINK1/Parkin or FUNDC1 |
CMA | Soluble proteins with KFERQ motif | Hsc70 / LAMP-2A |
Together, these autophagic pathways (Table 5) offer an orchestrated response that favors selective clearance and functional recovery rather than indiscriminate degradation, a hallmark of the hormetic action of singlet oxygen.
Autophagy pathway activation has been shown to prevent stress-induced tissue and organ injury. Beyond its immediate cytoprotective function, autophagy contributes to a wide range of systemic adaptations relevant to stress resilience, aging, immunity, and tissue homeostasis (Table 6).
Table 6 Systemic Resilience via the Autophagic Relay
System | Primary Target/Process | Hormetic Outcome |
Immune | Xenophagy & DAMP/PAMP regulation | Enhanced pathogen clearance; limited inflammasome activation. |
Nervous | Proteostasis (Tau/α-synuclein) | Neuroprotection; prevention of excitotoxicity. |
Skin | Keratinocyte/Fibroblast barrier repair | Accelerated wound healing; anti-aging (SIRT6-mediated). |
Cardiac | Mitophagy & Vascular NO signaling | Cardioprotection; preservation of vascular tone. |
Metabolic | Lipophagy & AT2 regeneration | Hepatic steatosis prevention; lung alveolar repair. |
Autophagy plays a multifaceted role in immune homeostasis. It mitigates chronic inflammation by degrading damaged organelles and oxidized macromolecules, thereby limiting the passive release of damage-associated molecular patterns (DAMPs)[lxiv] such as mitochondrial DNA, ATP, and oxidized proteins. This suppression of DAMP leakage helps prevent uncontrolled inflammasome activation and cytokine release. Conversely, under certain stress conditions, autophagy may also actively facilitate the secretion of select DAMPs, such as HMGB1, through non-classical pathways- initiating a context-dependent immunomodulatory feedback loop that can further enhance autophagy.[lxv]
In addition, autophagy functions as a key regulator of immune responses. It plays a crucial role in defending against intracellular pathogens by sequestering and degrading them through a process known as xenophagy,[lxvi] as well as contributing to antigen processing and presentation via MHC class II molecules, supports T cell development and homeostasis, and helps shape adaptive immune responses.
Through these mechanisms, autophagy serves as a multi-layered system that integrates microbial defense, inflammation control, and immune regulation, highlighting its potential as a therapeutic target for infectious, inflammatory, and autoimmune diseases.[lxvii]
In the aging context, basal autophagy levels decline, contributing to the accumulation of cellular damage. The age-related decline in basal autophagy is closely linked to the waning expression of the longevity-associated sirtuin, SIRT6. SIRT6 serves as a vital molecular switch that promotes autophagic flux by modulating the AMPK–mTOR signaling axis and increasing the expression of key autophagy-related genes (ATGs). Under conditions of mild oxidative stress, such as those induced by low-dose singlet oxygen, SIRT6 can be activated to facilitate the clearance of damaged organelles and proteotoxic aggregates that otherwise accumulate during senescence. By transiently restoring SIRT6-mediated autophagy, singlet oxygen may help ‘rejuvenate’ the cellular clearing apparatus, thereby preserving stem cell function and enhancing systemic resilience against age-related degeneration.67,69,[lxviii] Autophagy also preserves stem cell function across multiple tissues, including muscle, liver, and hematopoietic systems, by maintaining redox and mitochondrial integrity.[lxix]
Neuronal cells are especially vulnerable to oxidative damage due to their high metabolic demands and limited regenerative capacity. Autophagy helps remove dysfunctional mitochondria (mitophagy) and protein aggregates, maintaining proteostasis and preventing excitotoxicity. In the context of singlet oxygen exposure, selective autophagy helps prevent the accumulation of neurotoxic proteins such as α-synuclein and phosphorylated tau, processes implicated in neurodegenerative diseases[lxx],[lxxi] including Parkinson’s and Alzheimer’s[lxxii]. Moreover, autophagy contributes to neural development and synaptic plasticity, influencing learning and memory processes.106 Dysregulation of autophagy with aging or chronic oxidative stress can exacerbate excitotoxicity and synaptic loss, while controlled activation may confer neuroprotection.[lxxiii]
The skin can be viewed as a dynamic immuno-cutaneous ecosystem, where autophagy within skin cells plays a crucial role in preserving immune balance. [lxxiv] As the body’s largest interface with the environment, the skin is chronically exposed to environmental stressors such as ultraviolet (UV) radiation, pathogens, and oxidants, stressors that can damage keratinocytes, lipids, and extracellular matrix proteins. Autophagy plays a central role in removing harmful intracellular components, maintain cellular homeostasis, and prevent inflammation and premature aging.[lxxv]
A wide range of resident immune cells inhabit the skin, including Langerhans cells, dermal dendritic cells, macrophages, mast cells, and tissue-resident T cells. These immune cells are in ongoing communication with keratinocytes and other structural cells of the skin, collaboratively detecting and responding to pathogens, tumors, allergens, and autoantigens through intercellular signaling involving DAMPs and pathogen-associated molecular patterns (PAMPs) [lxxvi].
It has been shown that autophagy supports the function of key skin cells types, including keratinocytes, melanocytes, and immune cells, by regulating inflammation, promoting cell differentiation, and protecting against infection. In dermal fibroblasts, autophagy plays a critical role in modulating responses to DAMPs and PAMPs, limiting excessive immune activation through the degradation of damaged organelles and the regulation of cytokine release.113 In keratinocytes and epidermal stem cells, autophagy helps preserve skin renewal and barrier function, thereby reducing pathogen penetration and lowering the activation threshold of resident immune cells. In melanocytes, autophagy contributes to pigmentation homeostasis by regulating melanin distribution.[lxxx]
These functions highlight autophagy’s essential role in skin wound healing across all key phases: hemostasis, inflammation, proliferation, and remodeling.[lxxxi] It promotes keratinocyte proliferation and migration, drives fibroblast activation and differentiation, supports new blood vessel formation (angiogenesis), and helps clear damaged organelles and inflammatory mediators, supporting efficient tissue repair and re-epithelialization. Additionally, autophagy contributes to immune balance by regulating macrophage and neutrophil activity and supporting resolution of inflammation.
When autophagy is disrupted, it can lead to cellular senescence, chronic inflammation, impaired wound healing, and increased risk of pathological scarring or skin cancers. Dysregulated autophagy is also associated with immune-related skin disorders, including atopic dermatitis, psoriasis, and alopecia areata. 109
It can be concluded that the controlled induction of autophagy may offer a more effective, targeted approach for managing chronic skin conditions. It is also a promising therapeutic target for enhancing wound healing outcomes, treating skin aging and related disorders.
Autophagy plays a multifaceted role in lung injury and repair by supporting tissue-specific responses such as alveolar regeneration, redox homeostasis, and immune modulation. In particular, autophagy is essential for alveolar type 2 (AT2) progenitor cell–mediated regeneration following lung injury.[lxxxii] By promoting glucose, limiting lipid accumulation, and reducing oxidative stress, autophagy supports AT2 proliferation and epithelial barrier restoration.
In acute lung injury (ALI), autophagy interacts with the Nrf2 Nrf2 pathway to enhance antioxidant defense:[lxxxiii] by degrading the Nrf2 inhibitor Keap1, autophagy facilitates Nrf2 stabilization, contributing to protection against oxidative inflammation and preserving alveolar function. However, as in other tissues, the context and extent of autophagy determine its protective vs. detrimental effects
This balance becomes especially relevant in sepsis-induced lung injury, where autophagy maintains epithelial barrier integrity and regulates immune cell activity. Excessive or prolonged autophagic activation under systemic inflammatory stress may contribute to tissue damage and impaired resolution.[lxxxiv]
Autophagy also contributes to the pathogenesis of chronic pulmonary diseases:
Taken together, these findings underscore the context-dependent roles of autophagy in pulmonary health. Targeting autophagic signaling in a cell-type- and disease-specific manner may open new therapeutic avenues for both acute and chronic lung diseases.
Autophagy plays a critical role in maintaining cardiovascular health by supporting the turnover of damaged cellular components, particularly under stress conditions such as ischemia.[xc] In cardiac tissues, this process reduces myocardial injury, facilitates tissue remodeling, and promotes recovery after oxidative or mechanical insults. 139
When autophagy is defective or insufficient, cardiomyocytes accumulate dysfunctional mitochondria, misfolded proteins, and oxidative byproducts, [xci] which are hallmarks of cardiac aging and pathology. These impairments contribute to the progression of cardiomyopathies and heart failure. Mitophagy, the targeted removal of damaged mitochondria, is especially vital in the heart, where cells contain a high density of mitochondria and are constantly subjected to high oxidative and metabolic demands. By clearing depolarized mitochondria, mitophagy limits excessive ROS production and protects against ischemia–reperfusion injury. [xcii]
In the vascular system, autophagy also serves as a key homeostatic mechanism. Endothelial cells rely on basal autophagic activity to preserve nitric oxide signaling, limit vascular inflammation, and prevent atherogenesis. When this regulatory process is impaired, either by aging, chronic inflammation, or external oxidative stimuli such as singlet oxygen, vascular dysfunction and pro-atherogenic changes may ensue. 125
Importantly, the Nrf2–autophagy axis integrates redox-sensitive signaling with lipid metabolism and immune regulation, offering an additional layer of protection against oxidative and inflammatory stressors. Through this pathway, low-level singlet oxygen may promote adaptive remodeling and preserve vascular tone without triggering damaging oxidative overload. [xciii]
Together, these findings position autophagy as a central mechanism in cardiovascular resilience, mediating both cardiac and vascular adaptations to oxidative stress, including that induced by singlet oxygen exposure.
Autophagy maintains skeletal muscle homeostasis by removing dysfunctional proteins and organelles, particularly mitochondria, which is essential for preserving muscle mass, contractile performance, and metabolic adaptability. Mitophagy, the selective degradation of damaged mitochondria, plays a particularly critical role in muscle fibers, where high oxidative load and metabolic turnover necessitate constant mitochondrial quality control. Impairments in this process have been associated with various myopathies, including sarcopenia, muscular dystrophies, and cancer-associated cachexia, as accumulation of dysfunctional mitochondria and protein aggregates leads to fiber atrophy and impaired regeneration.[xciv],[xcv]
During physiological stress such as endurance exercise or caloric restriction, autophagy is transiently upregulated to promote muscle fiber remodeling, improve mitochondrial efficiency, and mobilize intracellular substrates for energy production. This dynamic regulation involves key redox-sensitive signaling hubs, including AMPK and the Nrf2 pathway, which coordinate the transcriptional and post-translational activation of autophagy machinery in response to oxidative fluctuations.[xcvi] Notably, exercise-induced production of singlet oxygen and other reactive oxygen species may act as a hormetic trigger to fine-tune autophagic activity, enhancing long-term muscle resilience and metabolic health. Understanding the precise balance of autophagic signaling in skeletal muscle may therefore hold therapeutic relevance for age-related muscle decline and metabolic syndromes.
Within endocrine tissues, autophagy plays a dual role in general cellular maintenance and in regulating hormone synthesis and secretion. In peptide-producing cells such as those of the pituitary gland, a specialized autophagic process known as crinophagy enables the degradation of excess secretory granules, thus preventing hormone hypersecretion and contributing to hormonal balance. [xcvii]
In steroidogenic cells of the adrenal cortex and testes, autophagy selectively targets mitochondria and smooth endoplasmic reticulum, which are organelles central to steroid biosynthesis. This regulatory role ensures metabolic efficiency and prevents oxidative damage, especially under stress conditions.[xcviii]
Additionally, autophagy in endocrine tissues is modulated by oxidative signals, including ROS such as singlet oxygen, which can trigger redox-sensitive autophagy pathways to adjust hormonal output. Dysfunctional autophagy in endocrine glands has been associated with various pathologies, including diabetes, infertility, and hormone-secreting tumors, due to disrupted secretory or steroidogenic functions.133,[xcix]
These findings suggest that targeted modulation of autophagy in endocrine tissues could represent a novel therapeutic approach to hormone-related disorders and metabolic diseases.
Autophagy plays a vital role in pregnancy by regulating trophoblast survival, supporting placental development, maintaining immune tolerance, and adapting to the dynamic metabolic demands of both mother and fetus. During early gestation, trophoblasts invade the uterine lining under low-oxygen conditions; here, autophagy facilitates cellular survival and promotes cytotrophoblast differentiation into the invasive phenotype essential for placental formation.
Autophagy also contributes to maternal–fetal immune tolerance by modulating inflammatory cytokine signaling, promoting tolerogenic dendritic cells, and regulating T cell activation. These effects reduce the risk of immune-mediated rejection of the semi-allogeneic fetus and help maintain immunological equilibrium at the maternal-fetal interface. [ci]
Additionally, under periods of maternal nutritional stress, autophagy enables intracellular recycling of macromolecules to maintain fetal nutrient supply and energy balance. Dysregulated autophagy, whether insufficient or excessive, has been implicated in various pregnancy complications, including preeclampsia, intrauterine growth restriction (IUGR), and gestational diabetes mellitus (GDM), highlighting its protective function in sustaining placental and fetal health. [cii]
The liver relies on autophagy for maintaining metabolic balance, detoxification, and redox homeostasis. 130,[ciii] Through lipophagy, autophagy facilitates the degradation of intracellular lipid droplets, contributing to lipid turnover and preventing hepatic steatosis. Simultaneously, mitophagy ensures the removal of dysfunctional mitochondria, protecting hepatocytes from oxidative stress and limiting ROS production. This mitochondrial quality control also supports energy-efficient regeneration of liver tissue following injury, ensuring adequate ATP production while minimizing oxidative damage.130,[civ] Dysregulation of hepatic autophagy has been implicated in liver diseases such as non-alcoholic fatty liver disease (NAFLD), alcoholic hepatitis, and fibrosis, underscoring its critical role in hepatocellular protection and regeneration.[cv]
Across a wide spectrum of tissues and physiological systems, autophagy emerges as a central regulatory process that integrates metabolic signals, oxidative cues, and stress adaptation. The evidence compiled in this chapter reveals a unifying theme: low-level oxidative stress, particularly that induced by singlet oxygen, can serve as a hormetic stimulus, activating autophagic pathways that support cellular renewal, immune modulation, and tissue repair (Figure 5).
In barrier tissues such as the skin and lungs, autophagy preserves epithelial integrity, controls inflammation, and enhances resilience to environmental insults. In skeletal and cardiac muscle, it maintains mitochondrial quality and metabolic homeostasis, particularly during physiological challenges like exercise or ischemia. In the liver, it coordinates lipid turnover and regeneration, while in the endocrine system and placenta, autophagy modulates hormonal synthesis and immune tolerance. Even during pregnancy, autophagy protects the maternal-fetal interface and contributes to placental development.
These diverse functions are governed by overlapping molecular mechanisms, including mitophagy, lipophagy, and redox-sensitive signaling via Nrf2, AMPK, and mTOR, yet are finely tuned to the needs of each tissue context. The dual role of autophagy in both cytoprotection and cell death is especially notable: the intensity and duration of singlet oxygen exposure may dictate whether autophagy serves a survival-promoting or pathological role, reflecting the delicate threshold between adaptive and maladaptive responses.
Understanding this context-dependence and threshold sensitivity is crucial for therapeutic modulation. Pharmacological or photo-induced activation of singlet oxygen may, under controlled conditions, be harnessed to stimulate beneficial autophagy, offering novel
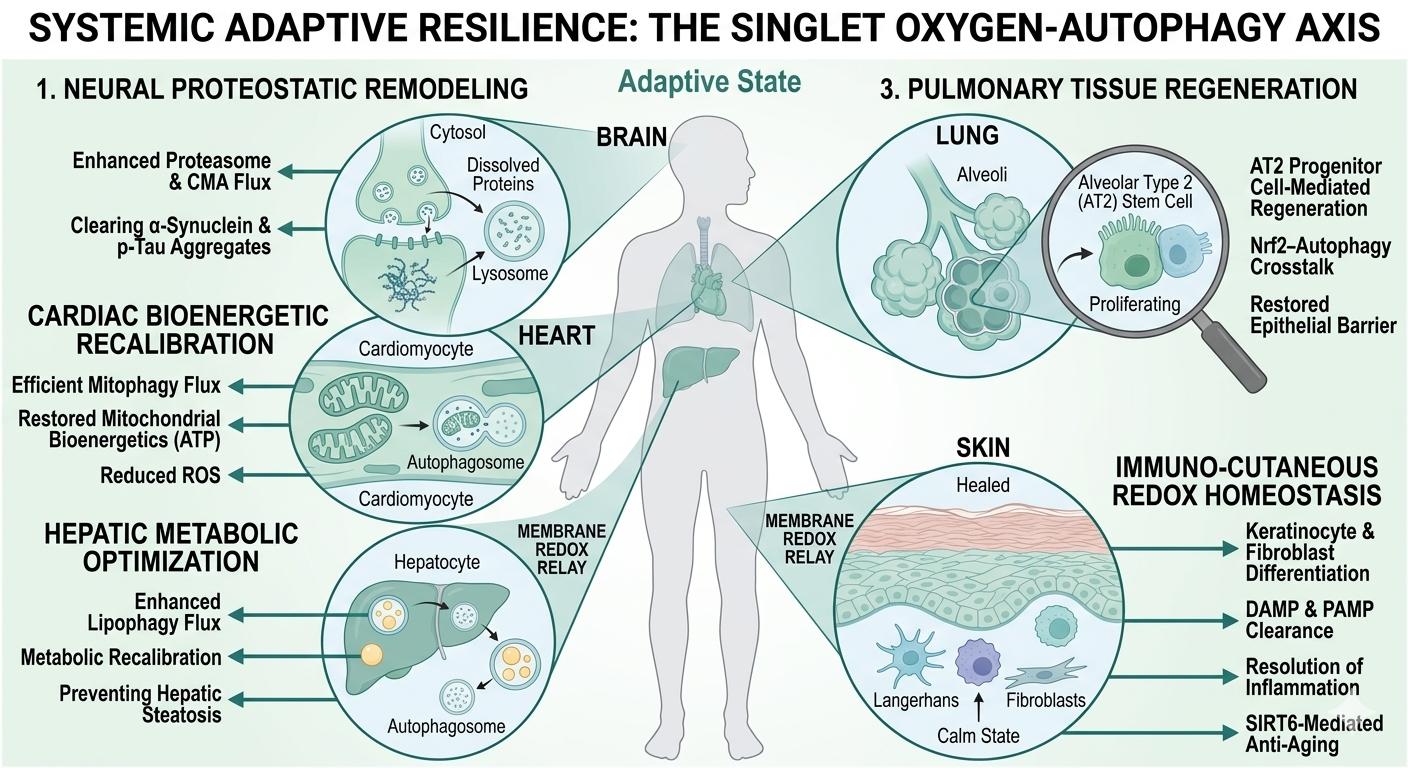
Figure 5 Schematic overview of the systemic adaptive landscape induced by the hormetic singlet oxygen-autophagy relay. The diagram illustrates the transition from a baseline state of cellular stagnation to a state of synchronized systemic recalibration. (1) Neural Proteostasis: Enhancement of autophagic and proteasomal flux facilitates the clearance of neurotoxic protein aggregates (e.g., α-synuclein, p-tau), preserving synaptic health. (2) Cardiac Bioenergetics: Targeted mitophagy eliminates dysfunctional, ROS-leaky mitochondria, restoring ATP production efficiency and myocardial resilience. (3) Pulmonary Regeneration: Activation of the Nrf2-autophagy crosstalk promotes the proliferation of Alveolar Type 2 (AT2) progenitor cells, essential for epithelial barrier restoration and adaptive remodeling. (4) Hepatic Metabolism: Induction of lipophagy optimizes lipid turnover and metabolic flux, mitigating hepatic steatosis and enhancing hepatocellular detoxification. (5) Immuno-Cutaneous Homeostasis: In the skin, the relay coordinates the clearance of DAMPs and PAMPs, facilitating SIRT6-mediated anti-aging effects and resolving chronic inflammatory signals within the dermal-epidermal ecosystem. Collectively, these tissue-specific responses demonstrate how a localized biophysical trigger at the plasma membrane is amplified into a body-wide program of functional rejuvenation and stress resistance. |
treatment avenues in inflammatory, degenerative, metabolic, and fibrotic diseases.
In summary, autophagy is not merely a stress response- it is a homeostatic amplifier of hormetic signaling. Through its ability to decode singlet oxygen cues into tissue-specific adaptive programs, autophagy bridges environmental sensing with intracellular resilience, affirming its relevance as both a biomarker and target of oxidative medicine.
[i] Gems, D., & Partridge, L. (2008). Stress-response hormesis and aging:“that which does not kill us makes us stronger”. Cell metabolism, 7(3), 200-203.
[ii] Shaw, P., & Chattopadhyay, A. (2020). Nrf2–ARE signaling in cellular protection: Mechanism of action and the regulatory mechanisms. Journal of Cellular Physiology, 235(4), 3119-3130.
[iii] Baird, L., Llères, D., Swift, S., & Dinkova-Kostova, A. T. (2013). Regulatory flexibility in the Nrf2-mediated stress response is conferred by conformational cycling of the Keap1-Nrf2 protein complex. Proceedings of the National Academy of Sciences, 110(38), 15259-15264.
[iv] Liu, X., Ren, S., Li, Z., Hao, D., Zhao, X., Zhang, Z., & Liu, D. (2023). Sirt6 mediates antioxidative functions by increasing Nrf2 abundance. Experimental cell research, 422(1), 113409.
[v] Rezazadeh, S., Yang, D., Tombline, G., Simon, M., Regan, S. P., Seluanov, A., & Gorbunova, V. (2019). SIRT6 promotes transcription of a subset of NRF2 targets by mono-ADP-ribosylating BAF170. Nucleic acids research, 47(15), 7914-7928.
[vi] Lu, S. C. (2013). Glutathione synthesis. Biochimica et Biophysica Acta (BBA)-General Subjects, 1830(5), 3143-3153.
[vii] Kensler, T. W., Wakabayashi, N., & Biswal, S. (2007). Cell survival responses to environmental stresses via the Keap1-Nrf2-ARE pathway. Annu. Rev. Pharmacol. Toxicol., 47(1), 89-116.
[viii] Conrad, M., & Pratt, D. A. (2019). The chemical basis of ferroptosis. Nature chemical biology, 15(12), 1137-1147.
[ix] Flohé, L., Toppo, S., & Orian, L. (2022). The glutathione peroxidase family: Discoveries and mechanism. Free Radical Biology and Medicine, 187, 113-122.
[x] Ma, Q. (2013). Role of nrf2 in oxidative stress and toxicity. Annual review of pharmacology and toxicology, 53(1), 401-426.
[xi] Grek, C. L., Zhang, J., Manevich, Y., Townsend, D. M., & Tew, K. D. (2013). Causes and consequences of cysteine S-glutathionylation. Journal of Biological Chemistry, 288(37), 26497-26504.
[xii] Telorack, M., Meyer, M., Ingold, I., Conrad, M., Bloch, W., & Werner, S. (2016). A glutathione-Nrf2-thioredoxin cross-talk ensures keratinocyte survival and efficient wound repair. PLoS genetics, 12(1), e1005800.
[xiii] (a) Ross, D., & Siegel, D. (2021). The diverse functionality of NQO1 and its roles in redox control. Redox Biology, 41, 101950. (b) Lee, W. S., Ham, W., & Kim, J. (2021). Roles of NAD (P) H: quinone oxidoreductase 1 in diverse diseases. Life, 11(12), 1301.
[xiv] (a) Waza, A. A., Hamid, Z., Ali, S., Bhat, S. A., & Bhat, M. A. (2018). A review on heme oxygenase-1 induction: is it a necessary evil. Inflammation Research, 67, 579-588. (b) Loboda, A., Damulewicz, M., Pyza, E., Jozkowicz, A., & Dulak, J. (2016). Role of Nrf2/HO-1 system in development, oxidative stress response and diseases: an evolutionarily conserved mechanism. Cellular and molecular life sciences, 73, 3221-3247. (c) Son, Y., Lee, J. H., Chung, H. T., & Pae, H. O. (2013). Therapeutic roles of heme oxygenase‐1 in metabolic diseases: curcumin and resveratrol analogues as possible inducers of heme oxygenase‐1. Oxidative medicine and cellular longevity, 2013(1), 639541.
[xv] Knoops, B., Argyropoulou, V., Becker, S., Ferté, L., & Kuznetsova, O. (2016). Multiple roles of peroxiredoxins in inflammation. Molecules and cells, 39(1), 60-64.
[xvi] Bafana, A., Dutt, S., Kumar, A., Kumar, S., & Ahuja, P. S. (2011). The basic and applied aspects of superoxide dismutase. Journal of Molecular Catalysis B: Enzymatic, 68(2), 129-138.
[xvii] Kirkman, H. N., & Gaetani, G. F. (2007). Mammalian catalase: a venerable enzyme with new mysteries. Trends in biochemical sciences, 32(1), 44-50.
[xviii] Lu, J., & Holmgren, A. (2014). The thioredoxin antioxidant system. Free radical biology and medicine, 66, 75-87.
[xix] Greeshma, M. V., Baidya, A., Mabalirajan, U., Madhunapantula, S. V., Thimmulappa, R. K., & Mahesh, P. A. (2024). Deciphering the role of 12/15-lipoxygenase in asthma: insights into mitochondrial dysfunction and therapeutic implications. Exploration of Asthma & Allergy, 2(6), 529-550.
[xx] Zhang, J., Zhang, M., Tatar, M., & Gong, R. (2025). Keap1-independent Nrf2 regulation: A novel therapeutic target for treating kidney disease. Redox Biology, 103593.
[xxi] Dong, H., Lyu, Y., Huang, C. Y., & Tsai, S. Y. (2025). Limiting cap-dependent translation increases 20S proteasomal degradation and protects the proteomic integrity in autophagy-deficient skeletal muscle. Autophagy, 21(6), 1212-1227.
[xxii] Kiang, J. G., & Tsokos, G. C. (1998). Heat shock protein 70 kDa: molecular biology, biochemistry, and physiology. Pharmacology & therapeutics, 80(2), 183-201.
[xxiii] Ameri, K., & Harris, A. L. (2008). Activating transcription factor 4. The international journal of biochemistry & cell biology, 40(1), 14-21.
[xxiv] (a) Richter-Landsberg, C., Wyttenbach, A., & Arrigo, A. P. (2009). The role of heat shock proteins during neurodegeneration in Alzheimer’s, Parkinson’s and Huntington’s disease. Heat shock proteins in neural cells, 81-99. (b) Zhang, N., Nao, J., Zhang, S., & Dong, X. (2024). Novel insights into the activating transcription factor 4 in Alzheimer’s disease and associated aging-related diseases: Mechanisms and therapeutic implications. Frontiers in Neuroendocrinology, 101144.
[xxv] (a) Bjørklund, G., Zou, L., Peana, M., Chasapis, C. T., Hangan, T., Lu, J., & Maes, M. (2022). The role of the thioredoxin system in brain diseases. Antioxidants, 11(11), 2161. (b) Cimini, A., Gentile, R., Angelucci, F., Benedetti, E., Pitari, G., Giordano, A., & Ippoliti, R. (2013). Neuroprotective effects of PrxI over‐expression in an in vitro human Alzheimer’s disease model. Journal of cellular biochemistry, 114(3), 708-715. (c) Zeng, X. S., Jia, J. J., Kwon, Y., Wang, S. D., & Bai, J. (2014). The role of thioredoxin-1 in suppression of endoplasmic reticulum stress in Parkinson disease. Free Radical Biology and Medicine, 67, 10-18.
[xxvi] (a) Ahsan, M. K., Lekli, I., Ray, D., Yodoi, J., & Das, D. K. (2009). Redox regulation of cell survival by the thioredoxin superfamily: an implication of redox gene therapy in the heart. Antioxidants & redox signaling, 11(11), 2741-2758. (b) Chong, C. R., Chan, W. P. A., Nguyen, T. H., Liu, S., Procter, N. E., Ngo, D. T., … & Horowitz, J. D. (2014). Thioredoxin-interacting protein: pathophysiology and emerging pharmacotherapeutics in cardiovascular disease and diabetes. Cardiovascular drugs and therapy, 28, 347-360.
[xxvii] Hao, X., Zhao, B., Towers, M., Liao, L., Monteiro, E. L., Xu, X., … & Zhang, R. (2024). TXNRD1 drives the innate immune response in senescent cells with implications for age-associated inflammation. Nature Aging, 4(2), 185-197.
[xxviii] Yang, Z. F., Drumea, K., Mott, S., Wang, J., & Rosmarin, A. G. (2014). GABP transcription factor (nuclear respiratory factor 2) is required for mitochondrial biogenesis. Molecular and cellular biology, 34(17), 3194-3201.
[xxix] (a) Gureev, A. P., Shaforostova, E. A., & Popov, V. N. (2019). Regulation of mitochondrial biogenesis as a way for active longevity: interaction between the Nrf2 and PGC-1α signaling pathways. Frontiers in genetics, 10, 435. (b) St-Pierre, J., Drori, S., Uldry, M., Silvaggi, J. M., Rhee, J., Jäger, S., … & Spiegelman, B. M. (2006). Suppression of reactive oxygen species and neurodegeneration by the PGC-1 transcriptional coactivators. Cell, 127(2), 397-408.
[xxx] Clements, C. M., McNally, R. S., Conti, B. J., Mak, T. W., & Ting, J. P. Y. (2006). DJ-1, a cancer-and Parkinson’s disease-associated protein, stabilizes the antioxidant transcriptional master regulator Nrf2. Proceedings of the National Academy of Sciences, 103(41), 15091-15096.
[xxxi] (a) Maiorino, M., Conrad, M., & Ursini, F. (2018). GPx4, lipid peroxidation, and cell death: discoveries, rediscoveries, and open issues. Antioxidants & redox signaling, 29(1), 61-74. (b) Lu, L., Oveson, B. C., Jo, Y. J., Lauer, T. W., Usui, S., Komeima, K., … & Campochiaro, P. A. (2009). Increased expression of glutathione peroxidase 4 strongly protects retina from oxidative damage. Antioxidants & redox signaling, 11(4), 715-724.
[xxxii] Roichman, A., Elhanati, S., Aon, M. A., Abramovich, I., Di Francesco, A., Shahar, Y., … & Cohen, H. Y. (2021). Restoration of energy homeostasis by SIRT6 extends healthy lifespan. Nature communications, 12(1), 3208.
[xxxiii] Cheng, J., Keuthan, C. J., & Esumi, N. (2023). The many faces of SIRT6 in the retina and retinal pigment epithelium. Frontiers in Cell and Developmental Biology, 11, 1244765.
[xxxiv] Palmeira, C. M., Teodoro, J. S., Amorim, J. A., Steegborn, C., Sinclair, D. A., & Rolo, A. P. (2019). Mitohormesis and metabolic health: The interplay between ROS, cAMP and sirtuins. Free Radical Biology and Medicine, 141, 483-491.
[xxxv] Kobayashi, E. H., Suzuki, T., Funayama, R., Nagashima, T., Hayashi, M., Sekine, H., … & Yamamoto, M. (2016). Nrf2 suppresses macrophage inflammatory response by blocking proinflammatory cytokine transcription. Nature communications, 7(1), 1-14.
[xxxvi] Guo, Z., Li, P., Ge, J., & Li, H. (2022). SIRT6 in aging, metabolism, inflammation and cardiovascular diseases. Aging and disease, 13(6), 1787.
[xxxvii] He, Y., Yang, G., Sun, L., Gao, H., Yao, F., Jin, Z., … & Lin, R. (2021). SIRT6 inhibits inflammatory response through regulation of NRF2 in vascular endothelial cells. International immunopharmacology, 99, 107926.
[xxxviii] Xu, W. D., Yang, C., & Huang, A. F. (2024). The role of Nrf2 in immune cells and inflammatory autoimmune diseases: a comprehensive review. Expert Opinion on Therapeutic Targets, 28(9), 789-806.
[xxxix] (a) Dodson, M., Shakya, A., Anandhan, A., Chen, J., Garcia, J. G., & Zhang, D. D. (2022). NRF2 and diabetes: the good, the bad, and the complex. Diabetes, 71(12), 2463-2476. (b) Vasileva, L. V., Savova, M. S., Amirova, K. M., Dinkova-Kostova, A. T., & Georgiev, M. I. (2020). Obesity and NRF2-mediated cytoprotection: Where is the missing link?. Pharmacological research, 156, 104760. (c) Li, S., Eguchi, N., Lau, H., & Ichii, H. (2020). The role of the Nrf2 signaling in obesity and insulin resistance. International journal of molecular sciences, 21(18), 6973.
[xl] (a) Zhang, Y., Wang, J., Wang, Y., & Lei, K. (2024). Nrf2/HO-1 signaling activation alleviates cigarette smoke-induced inflammation in chronic obstructive pulmonary disease by suppressing NLRP3-mediated pyroptosis. Journal of Cardiothoracic Surgery, 19(1), 58. (b) Audousset, C., McGovern, T., & Martin, J. G. (2021). Role of Nrf2 in disease: novel molecular mechanisms and therapeutic approaches–pulmonary disease/asthma. Frontiers in Physiology, 12, 727806.
[xli] Saha, S., Buttari, B., Profumo, E., Tucci, P., & Saso, L. (2022). A perspective on Nrf2 signaling pathway for neuroinflammation: a potential therapeutic target in Alzheimer’s and Parkinson’s diseases. Frontiers in cellular neuroscience, 15, 787258.
[xlii] Shi, T., & Dansen, T. B. (2020). Reactive oxygen species induced p53 activation: DNA damage, redox signaling, or both?. Antioxidants & Redox Signaling, 33(12), 839-859.
[xliii] Klotz, L. O., Briviba, K., & Sies, H. (1997). Singlet oxygen mediates the activation of JNK by UVA radiation in human skin fibroblasts. FEBS letters, 408(3), 289-291.
[xliv] Makino, T., Jinnin, M., Muchemwa, F. C., Fukushima, S., Kogushi‐Nishi, H., Moriya, C., … & Ihn, H. (2010). Basic fibroblast growth factor stimulates the proliferation of human dermal fibroblasts via the ERK1/2 and JNK pathways. British Journal of Dermatology, 162(4), 717-723.
[xlv] (a) Cuenda, A., & Rousseau, S. (2007). p38 MAP-kinases pathway regulation, function and role in human diseases. Biochimica et Biophysica Acta (BBA)-Molecular Cell Research, 1773(8), 1358-1375.
[xlvi] Klionsky, D. J. (2005). The molecular machinery of autophagy: unanswered questions. Journal of cell science, 118(Pt 1), 7. (b) Martindale, J. L., & Holbrook, N. J. (2002). Cellular response to oxidative stress: signaling for suicide and survival. Journal of cellular physiology, 192(1), 1-15.
[xlvii] (a) Wei, Y., Pattingre, S., Sinha, S., Bassik, M., & Levine, B. (2008). JNK1-mediated phosphorylation of Bcl-2 regulates starvation-induced autophagy. Molecular cell, 30(6), 678-688. (b) Yu, C., Minemoto, Y., Zhang, J., Liu, J., Tang, F., Bui, T. N., … & Lin, A. (2004). JNK suppresses apoptosis via phosphorylation of the proapoptotic Bcl-2 family protein BAD. Molecular cell, 13(3), 329-340.
[xlviii] (a) Zhou, J., Li, X. Y., Liu, Y. J., Feng, J., Wu, Y., Shen, H. M., & Lu, G. D. (2022). Full-coverage regulations of autophagy by ROS: from induction to maturation. Autophagy, 18(6), 1240-1255. (b) Przygoda, M., Bartusik-Aebisher, D., Dynarowicz, K., Cieślar, G., Kawczyk-Krupka, A., & Aebisher, D. (2023). Cellular mechanisms of singlet oxygen in photodynamic therapy. International Journal of Molecular Sciences, 24(23), 16890. (c) Liang, P., Kolodieznyi, D., Creeger, Y., Ballou, B., & Bruchez, M. P. (2020). Subcellular singlet oxygen and cell death: location matters. Frontiers in chemistry, 8, 592941.
[xlix] (a) Davies, K. J. (2000). Oxidative stress, antioxidant defenses, and damage removal, repair, and replacement systems. IUBMB life, 50(4‐5), 279-289. (b) Gao, Q. (2019). Oxidative stress and autophagy. Autophagy: biology and diseases: basic science, 179-198.
[l] (a) Kaminskyy, V. O., & Zhivotovsky, B. (2014). Free radicals in cross talk between autophagy and apoptosis. Antioxidants & redox signaling, 21(1), 86-102. (b) Yue, J., & López, J. M. (2020). Understanding MAPK signaling pathways in apoptosis. International journal of molecular sciences, 21(7), 2346.
[li] Kang, R., Zeh, H. J., Lotze, M. T., & Tang, D. J. C. D. (2011). The Beclin 1 network regulates autophagy and apoptosis. Cell Death & Differentiation, 18(4), 571-580.
[lii] Asgari, R., Yarani, R., Mohammadi, P., & Emami Aleagha, M. S. (2022). HIF-1α in the crosstalk between reactive oxygen species and autophagy process: a review in multiple sclerosis. Cellular and molecular neurobiology, 42(7), 2121-2129.
[liii] Romero, N. R., & Agostinis, P. (2014). Molecular mechanisms underlying the activation of autophagy pathways by reactive oxygen species and their relevance in cancer progression and therapy. In Autophagy: Cancer, Other Pathologies, Inflammation, Immunity, Infection, and Aging (pp. 159-178). Academic Press.
[liv] Horan, M. P., Pichaud, N., & Ballard, J. W. O. (2012). Quantifying mitochondrial dysfunction in complex diseases of aging. Journals of Gerontology Series A: Biomedical Sciences and Medical Sciences, 67(10), 1022-1035.
[lv] Osellame, L. D., Rahim, A. A., Hargreaves, I. P., Gegg, M. E., Richard-Londt, A., Brandner, S., … & Duchen, M. R. (2013). Mitochondria and quality control defects in a mouse model of Gaucher disease—links to Parkinson’s disease. Cell metabolism, 17(6), 941-953.
[lvi] Morciano, G., Patergnani, S., Bonora, M., Pedriali, G., Tarocco, A., Bouhamida, E., … & Pinton, P. (2020). Mitophagy in cardiovascular diseases. Journal of clinical medicine, 9(3), 892.
[lvii] Miao, M. Q., Han, Y. B., & Liu, L. (2023). Mitophagy in metabolic syndrome. The Journal of Clinical Hypertension, 25(5), 397-403.
[lviii] Park, J. H., Ko, J., Park, Y. S., Park, J., Hwang, J., & Koh, H. C. (2017). Clearance of damaged mitochondria through PINK1 stabilization by JNK and ERK MAPK signaling in chlorpyrifos-treated neuroblastoma cells. Molecular neurobiology, 54, 1844-1857.
[lix] (a) Chen, M., Chen, Z., Wang, Y., Tan, Z., Zhu, C., Li, Y., … & Chen, Q. (2016). Mitophagy receptor FUNDC1 regulates mitochondrial dynamics and mitophagy. Autophagy, 12(4), 689-702. (b) Park, S. Y., & Koh, H. C. (2020). FUNDC1 regulates receptor-mediated mitophagy independently of the PINK1/Parkin-dependent pathway in rotenone-treated SH-SY5Y cells. Food and Chemical Toxicology, 137, 111163. (c) He, H., Huang, W., Xiong, L., Ma, C., Wang, Y., Sun, P., … & Wu, Y. (2024). FUNDC1-mediated mitophagy regulates photodamage independently of the PINK1/Parkin-dependent pathway. Free Radical Biology and Medicine, 225, 630-640.
[lx] Majeski, A. E., & Dice, J. F. (2004). Mechanisms of chaperone-mediated autophagy. The international journal of biochemistry & cell biology, 36(12), 2435-2444.
[lxi] Xiong, Y., Contento, A. L., Nguyen, P. Q., & Bassham, D. C. (2007). Degradation of oxidized proteins by autophagy during oxidative stress in Arabidopsis. Plant physiology, 143(1), 291-299.
[lxii] Kiffin, R., Christian, C., Knecht, E., & Cuervo, A. M. (2004). Activation of chaperone-mediated autophagy during oxidative stress. Molecular biology of the cell, 15(11), 4829-4840.
[lxiii] Cuervo, A. M., & Dice, J. F. (2000). Unique properties of lamp2a compared to other lamp2 isoforms. Journal of cell science, 113(24), 4441-4450.
[lxiv] Zhang, Q., Kang, R., Zeh, III, H. J., Lotze, M. T., & Tang, D. (2013). DAMPs and autophagy: cellular adaptation to injury and unscheduled cell death. Autophagy, 9(4), 451-458.
[lxv] (a) Deretic, V. (2021). Autophagy in inflammation, infection, and immunometabolism. Immunity, 54(3), 437-453. (b) Tsuji, Y., Kuramochi, M., Golbar, H. M., Izawa, T., Kuwamura, M., & Yamate, J. (2020). Acetaminophen-induced rat hepatotoxicity based on M1/M2-macrophage polarization, in possible relation to damage-associated molecular patterns and autophagy. International Journal of Molecular Sciences, 21(23), 8998. (c) Levy, J. M. M., Towers, C. G., & Thorburn, A. (2017). Targeting autophagy in cancer. Nature Reviews Cancer, 17(9), 528-542.
[lxvi] Levine, B., Mizushima, N., & Virgin, H. W. (2011). Autophagy in immunity and inflammation. Nature, 469(7330), 323-335.
[lxvii] Deretic, V., Saitoh, T., & Akira, S. (2013). Autophagy in infection, inflammation and immunity. Nature Reviews Immunology, 13(10), 722-737.
[lxviii] Moore, M. N. (2020). Lysosomes, autophagy, and hormesis in cell physiology, pathology, and age-related disease. Dose-Response, 18(3), 1559325820934227.
[lxix] García-Prat, L., Martínez-Vicente, M., Perdiguero, E., Ortet, L., Rodríguez-Ubreva, J., Rebollo, E., … & Muñoz-Cánoves, P. (2016). Autophagy maintains stemness by preventing senescence. Nature, 529(7584), 37-42.
[lxx] Hara, T., Nakamura, K., Matsui, M., Yamamoto, A., Nakahara, Y., Suzuki-Migishima, R., … & Mizushima, N. (2006). Suppression of basal autophagy in neural cells causes neurodegenerative disease in mice. Nature, 441(7095), 885-889.
[lxxi] Zapata-Muñoz, J., Villarejo-Zori, B., Largo-Barrientos, P., & Boya, P. (2021). Towards a better understanding of the neuro-developmental role of autophagy in sickness and in health. Cell Stress, 5(7), 99.
[lxxii] Nixon, R. A. (2013). The role of autophagy in neurodegenerative disease. Nature medicine, 19(8), 983-997.
[lxxiii] Tomoda, T., Yang, K., & Sawa, A. (2020). Neuronal autophagy in synaptic functions and psychiatric disorders. Biological psychiatry, 87(9), 787-796.
[lxxiv] Sil, P., Wong, S. W., & Martinez, J. (2018). More than skin deep: autophagy is vital for skin barrier function. Frontiers in immunology, 9, 1376.
[lxxv] Jeong, D., Qomaladewi, N. P., Lee, J., Park, S. H., & Cho, J. Y. (2020). The role of autophagy in skin fibroblasts, keratinocytes, melanocytes, and epidermal stem cells. Journal of Investigative Dermatology, 140(9), 1691-1697.
[lxxvi] Richmond, J. M., & Harris, J. E. (2014). Immunology and skin in health and disease. Cold Spring Harbor perspectives in medicine, 4(12), a015339.
[lxxvii] Pivarcsi, A., Nagy, I., & Kemeny, L. (2005). Innate immunity in the skin: how keratinocytes fight against pathogens. Current immunology reviews, 1(1), 29-42.
[lxxviii] Davidson, S., Coles, M., Thomas, T., Kollias, G., Ludewig, B., Turley, S., … & Buckley, C. D. (2021). Fibroblasts as immune regulators in infection, inflammation and cancer. Nature Reviews Immunology, 21(11), 704-717.
[lxxix] Zhang, H., Xia, M., Li, H., Zeng, X., Jia, H., Zhang, W., & Zhou, J. (2025). Implication of Immunobiological Function of Melanocytes in Dermatology. Clinical Reviews in Allergy & Immunology, 68(1), 30.
[lxxx] Jeong, D., Qomaladewi, N. P., Lee, J., Park, S. H., & Cho, J. Y. (2020). The role of autophagy in skin fibroblasts, keratinocytes, melanocytes, and epidermal stem cells. Journal of Investigative Dermatology, 140(9), 1691-1697.
[lxxxi] Ren, H., Zhao, F., Zhang, Q., Huang, X., & Wang, Z. (2022). Autophagy and skin wound healing. Burns & trauma, 10, tkac003.
[lxxxii] Li, X., Wu, J., Sun, X., Wu, Q., Li, Y., Li, K., … & Chen, H. (2020). Autophagy reprograms alveolar progenitor cell metabolism in response to lung injury. Stem Cell Reports, 14(3), 420-432.
[lxxxiii] de la Vega, M. R., Dodson, M., Gross, C., Mansour, H. M., Lantz, R. C., Chapman, E., … & Zhang, D. D. (2016). Role of Nrf2 and autophagy in acute lung injury. Current pharmacology reports, 2, 91-101.
[lxxxiv] Yin, X., Xin, H., Mao, S., Wu, G., & Guo, L. (2019). The role of autophagy in sepsis: protection and injury to organs. Frontiers in physiology, 10, 1071.
[lxxxv] Barnes, P. J., Baker, J., & Donnelly, L. E. (2022). Autophagy in asthma and chronic obstructive pulmonary disease. Clinical Science, 136(10), 733-746.
[lxxxvi] Mizumura, K., Maruoka, S., Shimizu, T., & Gon, Y. (2018). Autophagy, selective autophagy, and necroptosis in COPD. International journal of chronic obstructive pulmonary disease, 3165-3172.
[lxxxvii] Araya, J., Kojima, J., Takasaka, N., Ito, S., Fujii, S., Hara, H., … & Kuwano, K. (2013). Insufficient autophagy in idiopathic pulmonary fibrosis. American Journal of Physiology-Lung Cellular and Molecular Physiology, 304(1), L56-L69.
[lxxxviii] Patel, A. S., Lin, L., Geyer, A., Haspel, J. A., An, C. H., Cao, J., … & Morse, D. (2012). Autophagy in idiopathic pulmonary fibrosis. PloS one, 7(7), e41394.
[lxxxix] McAlinden, K. D., Deshpande, D. A., Ghavami, S., Xenaki, D., Sohal, S. S., Oliver, B. G., … & Sharma, P. (2019). Autophagy activation in asthma airways remodeling. American journal of respiratory cell and molecular biology, 60(5), 541-553.
[xc] Liu, C., Liu, Y., Chen, H., Yang, X., Lu, C., Wang, L., & Lu, J. (2023). Myocardial injury: where inflammation and autophagy meet. Burns & Trauma, 11, tkac062.
[xci] Alcalai, R., Arad, M., Wakimoto, H., Yadin, D., Gorham, J., Wang, L., … & Seidman, C. E. (2021). LAMP2 cardiomyopathy: consequences of impaired autophagy in the heart. Journal of the American Heart Association, 10(17), e018829.
[xcii] Liu, Y., Wang, Y., Bi, Y., Zhao, Z., Wang, S., Lin, S., … & Mao, J. (2023). Emerging role of mitophagy in heart failure: from molecular mechanism to targeted therapy. Cell Cycle, 22(8), 906-918.
[xciii] Shen, Y., Liu, X., Shi, J., & Wu, X. (2019). Involvement of Nrf2 in myocardial ischemia and reperfusion injury. International journal of biological macromolecules, 125, 496-502.
[xciv] Chen, X., Ji, Y., Liu, R., Zhu, X., Wang, K., Yang, X., … & Sun, H. (2023). Mitochondrial dysfunction: roles in skeletal muscle atrophy. Journal of Translational Medicine, 21(1), 503.
[xcv] Li, P., Ma, Y., Yu, C., Wu, S., Wang, K., Yi, H., & Liang, W. (2021). Autophagy and aging: Roles in skeletal muscle, eye, brain and hepatic tissue. Frontiers in Cell and Developmental Biology, 9, 752962.
[xcvi] Ferraro, E., Giammarioli, A. M., Chiandotto, S., Spoletini, I., & Rosano, G. (2014). Exercise-induced skeletal muscle remodeling and metabolic adaptation: redox signaling and role of autophagy. Antioxidants & redox signaling, 21(1), 154-176.
[xcvii] Weckman, A., Di Ieva, A., Rotondo, F., Syro, L. V., Ortiz, L. D., Kovacs, K., & Cusimano, M. D. (2014). Autophagy in the endocrine glands. J Mol Endocrinol, 52(2), R151-R163.
[xcviii] (a) Afzal, A., Zhang, Y., Afzal, H., Saddozai, U. A. K., Zhang, L., Ji, X. Y., & Khawar, M. B. (2024). Functional role of autophagy in testicular and ovarian steroidogenesis. Frontiers in Cell and Developmental Biology, 12, 1384047. (b) Xu, R., Wang, F., Zhang, Z., Zhang, Y., Tang, Y., Bi, J., … & Tang, Z. (2023). Diabetes‐induced autophagy dysregulation engenders testicular impairment via oxidative stress. Oxidative medicine and cellular longevity, 2023(1), 4365895.
[xcix] (a) Kong, X., Wang, X., Xia, Q., Hu, Q., Yu, W., Huang, Q., … & Yu, J. (2025). Unveiling the nexus between environmental exposures and testicular damages: revelations from autophagy and oxidative stress imbalance. Cell Death Discovery, 11(1), 258. (b) Jung, H. S., & Lee, M. S. (2010). Role of autophagy in diabetes and mitochondria. Annals of the New York Academy of Sciences, 1201(1), 79-83.
[c] Zhao, X., Jiang, Y., Jiang, T., Han, X., Wang, Y., Chen, L., & Feng, X. (2020). Physiological and pathological regulation of autophagy in pregnancy. Archives of Gynecology and Obstetrics, 302(2), 293-303.
[ci] Chen, H., Chen, Y., & Zheng, Q. (2024). The regulated cell death at the maternal-fetal interface: beneficial or detrimental?. Cell Death Discovery, 10(1), 100.
[cii] Zhou, P., Wang, J., Wang, J., & Liu, X. (2024). When autophagy meets placenta development and pregnancy complications. Frontiers in Cell and Developmental Biology, 12, 1327167.
[ciii] Wang, Y., Singh, R., Xiang, Y., & Czaja, M. J. (2010). Macroautophagy and chaperone‐mediated autophagy are required for hepatocyte resistance to oxidant stress. Hepatology, 52(1), 266-277.
[civ] Xu, F., Hua, C., Tautenhahn, H. M., Dirsch, O., & Dahmen, U. (2020). The role of autophagy for the regeneration of the aging liver. International journal of molecular sciences, 21(10), 3606.
[cv] (a) Cursio, R., Colosetti, P., Codogno, P., Cuervo, A. M., & Shen, H. M. (2015). The role of autophagy in liver diseases: mechanisms and potential therapeutic targets. BioMed Research International, 2015, 480508. (b) Ruart, M., Chavarria, L., Campreciós, G., Suárez-Herrera, N., Montironi, C., Guixé-Muntet, S., … & Hernández-Gea, V. (2019). Impaired endothelial autophagy promotes liver fibrosis by aggravating the oxidative stress response during acute liver injury. Journal of hepatology, 70(3), 458-469.
The adaptations to oxidative stress unfold in a time-dependent manner84 and can result in long-lasting physiological benefits (Table 7). Early after oxidative insult, typically within minutes, NF-κB is rapidly activated, leading to a transient pro-inflammatory response that primes the system for defense and cellular recruitment.[i] Within 1–3 hours, these early signals stimulate transcriptional programs, notably via Nrf2, promoting transcription of phase II detoxification enzymes and antioxidant defenses, such as HO-1 and NQO1. As these protective proteins accumulate, they provide an enhanced state of stress resilience lasting up to 48 hours. 84,[ii],[iii]
During this intermediate phase, lipid peroxidation by-products like 4-hydroxynonenal (4-HNE) act as secondary messengers that stably modify proteins and influence cell signaling well beyond the initial oxidative event.142 This extended adaptive phase is further supported by the induction of autophagy, typically induced several hours after exposure, contributing to cellular maintenance by clearing damaged organelles.141 These processes are complemented by cellular repair mechanisms, activated to restore redox balance and tissue integrity.
Together, these temporally orchestrated responses to moderate oxidative stress, such as that induced by singlet oxygen, foster a sustained hormetic effect that contributes to cellular preconditioning, enhanced metabolic regulation, and long-term resilience to stress.
Table 7 Timing and duration of defense responses to moderate oxidative stress
Response Type | Trigger | Onset (Approx.) | Duration |
NF-κB activation | ROS | Minutes | 1–3 hours |
Nrf2 activation | 4-HNE, ROS | 1–3 hours | 6–48 hours (longer term) |
Autophagy induction | Persistent ROS | Several hours | Long-lasting (days) |
Cellular repair | Nrf2/ARE genes | After Nrf2 signal | Hours–days |
[i] Jaganjac, M., Milkovic, L., Zarkovic, N., & Zarkovic, K. (2022). Oxidative stress and regeneration. Free Radical Biology and Medicine, 181, 154-165.
[ii] Franzini, M., Valdenassi, L., Pandolfi, S., Tirelli, U., Ricevuti, G., & Chirumbolo, S. (2023). The role of ozone as an Nrf2-keap1-ARE activator in the anti-microbial activity and immunity modulation of infected wounds. Antioxidants, 12(11), 1985.
[iii] Clavo, B., Rodríguez-Esparragón, F., Rodríguez-Abreu, D., Martínez-Sánchez, G., Llontop, P., Aguiar-Bujanda, D., … & Santana-Rodríguez, N. (2019). Modulation of oxidative stress by ozone therapy in the prevention and treatment of chemotherapy-induced toxicity: review and prospects. Antioxidants, 8(12), 588.
The transition from cellular signaling to systemic health is mediated by the sustained adaptive responses described previously. By maintaining high levels of cellular integrity and preventing premature apoptosis, low-dose singlet oxygen supports an extended healthspan and enhanced resistance to age-related degeneration.[i] These benefits manifest across several complex physiological systems:
[i] (a) Ristow, M., & Zarse, K. (2010). How increased oxidative stress promotes longevity and metabolic health: The concept of mitochondrial hormesis (mitohormesis). Experimental gerontology, 45(6), 410-418. (b) Calabrese, E. J., Osakabe, N., Di Paola, R., Siracusa, R., Fusco, R., D’Amico, R., … & Calabrese, V. (2023). Hormesis defines the limits of lifespan. Ageing research reviews, 102074.
[ii] Hulten, L. M., Holmström, M., & Soussi, B. (1999). Harmful singlet oxygen can be helpful. Free Radical Biology and Medicine, 27(11), 1203-1207.
[iii] (a) Bieber, K., Hundt, J. E., Yu, X., Ehlers, M., Petersen, F., Karsten, C. M., … & Ludwig, R. J. (2023). Autoimmune pre-disease. Autoimmunity Reviews, 22(2), 103236. (b) Xiao, Z. X., Miller, J. S., & Zheng, S. G. (2021). An updated advance of autoantibodies in autoimmune diseases. Autoimmunity Reviews, 20(2), 102743.
[iv] (a) Tisoncik, J. R., Korth, M. J., Simmons, C. P., Farrar, J., Martin, T. R., & Katze, M. G. (2012). Into the eye of the cytokine storm. Microbiology and molecular biology reviews, 76(1), 16-32. (b) Karki, R., & Kanneganti, T. D. (2021). The ‘cytokine storm’: Molecular mechanisms and therapeutic prospects. Trends in immunology, 42(8), 681-705.
Given the short lifetime and high reactivity of singlet oxygen, it is unlikely that singlet oxygen itself serves as a long-range traveling messenger within the body. Rather, singlet oxygen is best conceptualized as a localized “biophysical trigger” that initiates secondary messenger cascades capable of propagation through blood, lymphoid trafficking, extracellular vesicle transfer, and neuroimmune reflex arcs. By coordinating these diverse pathways, a localized surface exposure is translated into a widespread, systemic adaptive response.
The therapeutic effects of low-level singlet oxygen-enriched air are initiated primarily through contact with the body’s exposed external surfaces,20 most notably, the skin, respiratory epithelium, sclera, and reproductive tract mucosa. These surfaces act as dynamic biochemical sensors rather than passive barriers.
At the forefront of this sensing are redox-sensitive biomolecules embedded in the plasma membranes of epithelial and immune cells. Upon exposure to singlet oxygen, specific lipids and thiol-containing membrane proteins undergo oxidative modification, which initiates intracellular signalling cascades that regulate gene expression related to antioxidant defenses, inflammation, and cellular repair. 84,[i] While resident immune cells are vital mediators, the propagation of the hormetic signal relies on an integrated network of communication involving several pathways:
While singlet oxygen acts as the initial biophysical trigger, the propagation of its effects beyond the point of contact is driven by the secretion of signaling molecules and membrane-bound packages.
While the skin’s immune role is detailed in section 5.3.3.3, it is important to note that similar redox-sensing and signaling occurs in respiratory and ocular surfaces. Resident immune cells, particularly macrophages in the lungs[iv] and conjunctiva[v], are responsive to these oxidative cues. These cells not only engage in local tissue maintenance and immune surveillance but also serve as mobile mediators of systemic communication.
Once activated by low-level oxidative stress, macrophages may enter the circulatory system and exert systemic effects, including the regulation of hematopoiesis (the formation of new blood cells) and modulation of inflammation in distant tissues. In the lungs, macrophages help maintain tissue homeostasis during oxidative stress by releasing antioxidant enzymes, engulfing and removing damaged cells, and promoting tissue repair through secretion of growth factors that stimulate epithelial cell proliferation and matrix metalloproteinases (MMPs), which facilitate the breakdown and remodeling of damaged extracellular matrix components. [vi]
As oxidative stress often triggers pro-inflammatory signaling,[vii] macrophages play a key role in regulating the immune response by releasing anti-inflammatory cytokines that counteract these effects. They also secrete cytokines and chemokines that recruit other immune cells, such as T cells and B cells, to sites of injury, thereby shaping the adaptive immune response, promoting immune tolerance, and supporting the restoration of tissue homeostasis. [viii]
Macrophages possess the capacity to migrate throughout the body in response to diverse physiological and pathological signals.[ix] During systemic inflammation or disease states, lung-resident macrophages may enter the bloodstream and localize to sites of injury or inflammation, where they contribute to immune regulation and tissue repair. Some may also migrate to the bone marrow, where they influence hematopoiesis and help maintain macrophage populations in organs such as the spleen, liver, and peritoneal cavity.[x]
This migratory behavior is tightly orchestrated by chemokines, cytokines, and other signaling molecules that direct immune cell trafficking and tissue-specific homing.157 Through this systemic mobility, lung-derived macrophages extend their influence beyond the pulmonary environment, participating in immune surveillance, defense, and the resolution of inflammation throughout the body. These mechanisms support the broader hypothesis that local singlet oxygen exposure may translate into widespread immunomodulatory benefits.
Like pulmonary macrophages, skin and conjunctival macrophages are strategically positioned at external interfaces and respond dynamically to oxidative stress. Upon activation, they release antioxidant enzymes and inflammatory mediators, migrate to lymphoid tissues, or emit systemic signals,[xi] contributing to both local defense and broader immune modulation. These properties reinforce the plausibility of systemic effects resulting from surface-level singlet oxygen exposure.
Because singlet oxygen is a potent “biophysical trigger,” it can directly modulate sensory nerve endings at the body’s external interfaces (skin and respiratory tract). This initiates a rapid electrical-to-chemical signaling loop that bypasses the need for physical molecule transport.
The neural propagation of hormesis involves specific molecular switches that bridge the gap between electrical signals and cellular gene expression:
The multi-channel propagation of the singlet oxygen signal, facilitated by epithelial-vesicular relays, migratory immune cells, and rapid neuroimmune reflex arcs, provides a robust biological framework for the systemic benefits observed in low-dose therapy. This integrated network explains how a short-lived, gas-phase molecule acting on a primary interface (such as the lungs or skin) can induce a state of “systemic hormesis.” By simultaneously activating genetic survival pathways (Nrf2/ARE) and calibrating the inflammatory tone via neural and immune signaling, the body transitions into a state of heightened resilience and accelerated repair.
This theoretical foundation is essential for interpreting the clinical and observational data that follow. Whether addressing localized tissue damage or chronic systemic imbalances, the efficacy of singlet oxygen-enriched air relies on this seamless translation of a surface-level biophysical cue into a coordinated physiological response. In the following section, we examine how these mechanisms manifest in specific clinical contexts, ranging from respiratory and dermatological recovery to systemic metabolic and cognitive improvements.
[i] Fridovich, I. (2013). Oxygen: how do we stand it?. Medical Principles and Practice, 22(2), 131-137.
[ii] (a) Meyer, J. G., Garcia, T. Y., Schilling, B., Gibson, B. W., & Lamba, D. A. (2019). Proteome and secretome dynamics of human retinal pigment epithelium in response to reactive oxygen species. Scientific reports, 9(1), 15440. (b) Yi, Y., Wang, X. R., Chen, H. T., Huang, W. Y., Feng, L. X., Fang, S. B., & Xiong, G. X. (2023). Development of a serum-free culture method for endothelial cells of the stria vascularis and their pro-inflammatory secretome changes induced by oxidative stress. Clinical and Experimental Otorhinolaryngology, 16(1), 37-48.
[iii] Eldh, M., Ekström, K., Valadi, H., Sjöstrand, M., Olsson, B., Jernås, M., & Lötvall, J. (2010). Exosomes communicate protective messages during oxidative stress; possible role of exosomal shuttle RNA. PloS one, 5(12), e15353.
[iv] (a) Evren, E., Ringqvist, E., Tripathi, K. P., Sleiers, N., Rives, I. C., Alisjahbana, A., … & Willinger, T. (2021). Distinct developmental pathways from blood monocytes generate human lung macrophage diversity. Immunity, 54(2), 259-275. (b) Tan, S. Y., & Krasnow, M. A. (2016). Developmental origin of lung macrophage diversity. Development, 143(8), 1318-1327.
[v] Chinnery, H. R., McMenamin, P. G., & Dando, S. J. (2017). Macrophage physiology in the eye. Pflügers Archiv-European Journal of Physiology, 469, 501-515.
[vi] Puttur, F., Gregory, L. G., & Lloyd, C. M. (2019). Airway macrophages as the guardians of tissue repair in the lung. Immunology and cell biology, 97(3), 246-257.
[vii] Rahman, I., & Adcock, I. M. (2006). Oxidative stress and redox regulation of lung inflammation in COPD. European respiratory journal, 28(1), 219-242.
[viii] Arora, S., Dev, K., Agarwal, B., Das, P., & Syed, M. A. (2018). Macrophages: Their role, activation and polarization in pulmonary diseases. Immunobiology, 223(4-5), 383-396.
[ix] Ogura, M., & Kitamura, M. (1998). Oxidant stress incites spreading of macrophages via extracellular signal-regulated kinases and p38 mitogen-activated protein kinase. The Journal of Immunology, 161(7), 3569-3574.
[x] Reichard, A., Wanner, N., Farha, S., & Asosingh, K. (2023). Hematopoietic stem cells and extramedullary hematopoiesis in the lungs. Cytometry Part A, 103(12), 967-977.
[xi] (a) Lämmermann, T., & Sixt, M. (2008). The microanatomy of T‐cell responses. Immunological reviews, 221(1), 26-43. (b) Chinnery, H. R., McMenamin, P. G., & Dando, S. J. (2017). Macrophage physiology in the eye. Pflügers Archiv-European Journal of Physiology, 469, 501-515.
[xii] (a) Andersson, D. A., Gentry, C., Moss, S., & Bevan, S. (2008). Transient receptor potential A1 is a sensory receptor for multiple products of oxidative stress. Journal of Neuroscience, 28(10), 2485-2494. (b) Kozai, D., Ogawa, N., & Mori, Y. (2014). Redox regulation of transient receptor potential channels. Antioxidants & redox signaling, 21(6), 971-986.
[xiii] (a) Tracey, K. J. (2002). The inflammatory reflex. Nature, 420(6917), 853-859. (b) Pavlov, V. A., & Tracey, K. J. (2017). Neural regulation of immunity: molecular mechanisms and clinical translation. Nature neuroscience, 20(2), 156-166.
[xiv] (a) Antunes, G. L., Silveira, J. S., Kaiber, D. B., Luft, C., da Costa, M. S., Marques, E. P., … & da Cunha, A. A. (2020). Cholinergic anti‐inflammatory pathway confers airway protection against oxidative damage and attenuates inflammation in an allergic asthma model. Journal of Cellular Physiology, 235(2), 1838-1849. (b) Kelly, M. J., Breathnach, C., Tracey, K. J., & Donnelly, S. C. (2022). Manipulation of the inflammatory reflex as a therapeutic strategy. Cell Reports Medicine, 3(7).
[xv] Kozai, D., Ogawa, N., & Mori, Y. (2014). Redox regulation of transient receptor potential channels. Antioxidants & redox signaling, 21(6), 971-986.
[xvi] Gupta, A. K., Gupta, S., Mehan, S., Khan, Z., Das Gupta, G., & Narula, A. S. (2025). Exploring the connection between BDNF/TrkB and AC/cAMP/PKA/CREB signaling pathways: potential for neuroprotection and therapeutic targets for neurological disorders. Molecular Neurobiology, 62(11), 14627-14659.
[xvii] Bellavance, M. A., & Rivest, S. (2014). The HPA–immune axis and the immunomodulatory actions of glucocorticoids in the brain. Frontiers in immunology, 5, 136.
Given the systemic reach described above, the potential applications of low-dose singlet oxygen technology span a wide array of physiological systems. The following sections detail the evidence for its role as a supportive adjunct in conventional medical protocols and its impact on recovery, performance, and chronic health management.
This section presents a summary of selected individual patient reports and clinical observations describing subjective or objective functional improvements following low-dose Singlet Oxygen Therapy (SOT), administered via a patented device that generates low-dose singlet oxygen without irradiation.18
Given that the respiratory epithelium is the most direct interface for gas-phase singlet oxygen, the pulmonary system serves as a primary site for both local repair and the initiation of systemic signals.
Asthma is a chronic inflammatory airway disease characterized by bronchial hyperresponsiveness, reversible airway obstruction, and recurrent symptoms such as wheezing, coughing, and breathlessness. Common triggers include allergens, exercise, infections, and environmental irritants. During exacerbations, airway inflammation leads to bronchial constriction, edema, and mucus production, reducing airflow and impairing gas exchange.[i]
Repeated asthma attacks increase the risk of airway remodeling and irreversible airflow limitation. Conventional therapies, including corticosteroids and bronchodilators, are effective but often carry side effects, and many patients remain symptomatic despite optimized pharmacological management.[ii] Therefore, there is a growing interest in adjunctive therapies aimed at reducing attack frequency, minimizing reliance on synthetic drugs, and enhancing endogenous healing responses.
Observational Reports of SOT Use in Asthma
Self-reported observations suggest that singlet oxygen-enriched air may confer both acute relief and longer-term benefits for asthma patients. Users have described rapid alleviation of breathlessness during attacks, as well as a gradual reduction in attack frequency and medication dependence. Notably, several individuals who previously required prophylactic steroids for seasonal asthma reported discontinuing their medication after consistent nightly SOT. Some also reported improvements in symptoms associated with airway remodeling.
Preliminary Clinical Study
In a small observational study conducted in Spain by Prof. M. Fegricio,[iii] two groups of 25 asthma patients were evaluated over 30 and 60 days. The treatment group used a low-level ambient singlet oxygen generator daily, while the control group received a sham device. Patient-reported outcomes (Figure 6) showed significant improvements in sleep quality and energy in the treatment group, alongside a reduction in weekly asthma attacks (from 1.08 to 0.48 attacks/week). Sixteen patients discontinued all pharmacologic treatment by the study’s end, while no such changes occurred in the control group.
Figure 6 Change in the average rating of energy levels and sleep quality of 25 asthma patients, with (left graph) and without (right graph) daily usage of low-level ambient singlet oxygen generator over a period of 60 days.Error! Bookmark not defined.
Proposed Mechanism: While preliminary and based on observational data, these findings are consistent with the mechanistic rationale discussed in Sections 5 and 7. Specifically, the controlled oxidative signal generated by singlet oxygen may activate redox-sensitive anti-inflammatory and epithelial repair pathways. These processes could improve airway tone and mucosal integrity, thereby lowering susceptibility to bronchoconstriction. However, controlled trials are essential to validate these effects and rule out placebo influences or seasonal fluctuations.
Chronic obstructive pulmonary disease (COPD), a progressive respiratory disorder encompassing chronic bronchitis and emphysema, is characterized by persistent airflow limitation, mucus overproduction, and alveolar destruction. As the third leading cause of death in the United States,[iv] COPD currently has no cure, only supportive treatments are available, aimed at slowing disease progression and managing symptoms.
Patients with advanced COPD often experience debilitating breathlessness, frequent exacerbations, and, eventually, dependence on supplemental oxygen. Without a lung transplant, this condition ultimately proves fatal. In this context, low-level singlet oxygen exposure has been anecdotally associated with perceived improvements in breathing and function.
Several users have reported enhanced respiratory ease and a reduction in oxygen dependence within days of initiating therapy. A few individuals with advanced disease described regaining the ability to work and travel, improvements that warrant systematic investigation. In some cases, spirometry conducted after a month of use revealed increases in forced expiratory volume (FEV₁) and improved FEV₁/FVC ratios, outcomes not commonly observed in late-stage COPD with non-invasive methods.
These preliminary findings align with earlier observational reports, such as a small, unpublished study in which patients with moderate-to-severe COPD underwent a four-week regimen of singlet oxygen inhalation therapy.[v] Participants demonstrated modest improvements in FEV₁, peak expiratory flow, and a reduction in symptom burden and bronchodilator use. Although the study lacked a control group and peer review, its outcomes parallel known physiological mechanisms described in Sections 5 and 7, including redox-modulated inflammation control, epithelial repair, and enhanced mitochondrial turnover.
The following case observations provide individual examples of patients with moderate to severe COPD who reported respiratory improvements following routine use of SOT.
Case 1: Functional and Spirometric Improvement in a Patient with Severe COPD Following SOT
A 67-year-old male with a 20-year smoking history (approx. 1 pack/day) was diagnosed with severe COPD. Despite smoking cessation, his respiratory condition progressively worsened, with persistent wheezing, dyspnea, and pulmonary congestion that impaired basic activities such as walking and showering.
Initial treatment included corticosteroids (prednisone) and antibiotics (cephalexin), though his lung function continued to deteriorate. FEV₁ declined from 52% to 35% predicted within a year. At this point, physicians recommended long-term oxygen therapy and pulmonary rehabilitation.
In April 2017, the patient initiated daily SOT. Within days, he reported subjective improvement in breathing and energy levels. Over the following weeks, he resumed travel and returned to work.
Spirometry one month into therapy indicated an increase in FEV₁ from 35% to 58% predicted, with further improvement (63%) over the next year. No changes in standard medications were recorded during this time.
Case 2: Remission of COPD With Discontinuation of Inhaled Therapy Following SOT
A 57-year-old female with heavy smoking history was diagnosed in 2020 with moderate COPD, confirmed by spirometry (FEV₁ ~83%; FEV₁/FVC <80%). She experienced progressive dyspnea, wheezing, and exertional fatigue that interfered with her occupation as a children’s entertainer.
In 2021, the patient began nightly SOT. During the first week, she experienced productive coughing with green sputum, possibly reflecting a mucolytic or detoxification response. Over the next two months, she noted substantial improvements in breathing and physical stamina. Wheezing fully resolved within three months.
Initially prescribed four inhaled medications, including corticosteroids and bronchodilators, the patient gradually discontinued them over a six-month period. By year’s end, she remained asymptomatic and off all inhalers.
Pulmonary function tests in 2023 and 2025 showed normalized spirometry (FEV₁ >95%; FEV₁/FVC ≥83%). A pulmonologist concluded that the patient no longer met diagnostic criteria for COPD.
The patient has since resumed working and other physical activities, including running and long walks, without any recurrence of respiratory symptoms. She currently reports full respiratory health.
Proposed Mechanism: These cases represent only 2 examples of multiple cases in which users with COPD have experienced long-term COPD remission, both in symptoms and lung function, following SOT, suggesting that singlet oxygen-enriched air may assist in reducing the oxidative burden in the lungs. By triggering the Nrf2-mediated antioxidant response within alveolar macrophages and epithelial cells, the therapy helps neutralize the chronic ROS production characteristic of COPD. Beyond inflammation, the activation of neuroimmune reflex arcs and the potential modulation of nitric oxide (NO) bioavailability may contribute to improved capillary dilation and more efficient oxygen exchange.
In addition to its profound clinical burden, COPD imposes a substantial financial toll. An analysis done in 2020[vi] estimated that respiratory diseases cost the U.S. healthcare system $170.8 billion annually. Asthma accounted for the highest costs, followed by COPD. For asthma, prescription drugs represented the largest share (48%), while for COPD, hospitalizations (28.8%) and prescriptions (28.5%) were primary cost drivers.
Most COPD-related spending was concentrated in adults over 45, with nearly 70% covered by public insurers. In contrast, asthma-related costs were more evenly distributed across age groups.
These data highlight the potential value of adjunct therapies that can reduce exacerbation rates, hospitalizations, and medication use. If low-dose SOT is validated in future clinical studies, it may represent a cost-effective strategy that leverages endogenous repair pathways rather than relying solely on pharmacological suppression.
Pneumonia remains a significant global health concern, affecting over 500 million individuals annually and accounting for approximately 4 million deaths, which is about 7% of all global mortality. It is the leading infectious cause of death in children under five, particularly in regions with limited access to medical care, such as South Asia and sub-Saharan Africa.[vii]
The condition arises when an infection, which can be bacterial, viral, or fungal, triggers inflammation in the lungs, leading to alveolar fluid accumulation, impaired gas exchange, and respiratory distress. Diagnosis and treatment are often complicated by uncertainty around the causative pathogen and the rise in antimicrobial resistance, which can hinder effective, targeted therapy.
Observational Reports of SOT Use in Pneumonia
Several users of singlet oxygen therapy (SOT) have anecdotally reported significant symptomatic relief within 1–3 days of starting therapy. These effects included:
Some individuals also noted a shortened recovery timeline, which is typically protracted in pneumonia, often lasting several weeks.
Proposed Mechanism: The reported improvements in respiratory function and recovery following singlet oxygen therapy may be explained by its capacity to modulate redox-sensitive biological pathways. Low-dose oxidative stimulation is known to reduce local inflammation, promote epithelial repair, and enhance mucociliary clearance, key processes in the resolution of pneumonia. Additionally, by supporting immune regulation and restoring redox balance in the lung microenvironment, singlet oxygen may facilitate more efficient pathogen clearance and tissue recovery. These mechanisms, detailed in Section 5, offer a biologically plausible framework for understanding how SOT could accelerate symptom resolution and shorten recovery time in individuals with pneumonia.
Excessive phlegm production is a common and distressing symptom across a broad spectrum of respiratory conditions, including asthma, [viii] COPD,[ix] pneumonia,[x] allergic rhinitis,[xi] chronic exposure to tobacco smoke[xii] and other conditions[xiii]. While mucus is essential for trapping pathogens and particulates,[xiv] abnormal accumulation or viscosity can impair airflow, reduce oxygenation, and heighten infection risk. In severe cases, it contributes to hypoxemia, atelectasis, and respiratory failure.[xv]
Conventional therapies, which include expectorants, mucolytics, bronchodilators, and physical airway clearance techniques, often offer limited relief and may be impractical or burdensome, especially in patients with reduced mobility or chronic illness. Moreover, these treatments do not address upstream dysregulation in inflammatory or redox pathways that often underlie mucus hypersecretion.
Reports on Mucus Reduction Following SOT
Following SOT, users have consistently reported improvements in mucus-related symptoms, including reductions in phlegm volume and viscosity. These reports span a variety of respiratory conditions, including asthma, COPD, pneumonia, allergies, and chronic smoking-related bronchitis. Notably, some individuals described the expulsion of thick, dark mucus during early exposure, interpreted as a “detoxification phase,” followed by an enduring sense of airway clearance and improved breathing.
Case: Discontinuation of Airway Suctioning in a Non-Communicative Patient
non-communicative adult with a severe brain injury required routine nightly suctioning to prevent aspiration due to excessive phlegm production. Following SOT, the need for suctioning diminished and ultimately ceased. His mother and caregiver reported improved sleep and overall well-being for both herself and the patient.
Proposed Mechanism: These effects are consistent with redox-modulatory mechanisms described in Section 5. Low-dose singlet oxygen likely reduces local airway inflammation, downregulating the inflammatory pathways that drive excess mucus production.[xvi] It may also stimulate epithelial repair117 and enhance ciliary activity[xvii], both of which are crucial for effective mucociliary clearance. Additionally, redox-sensitive modulation of mucus viscosity may lower its viscosity, facilitating easier expectoration.[xviii]
Taken together, these effects suggest that singlet oxygen may help restore the mucus production-clearance cycle, particularly in individuals with impaired mucociliary function or chronic inflammation. As a non-invasive, low-burden approach, this therapy could complement existing treatments or offer relief to patients with refractory symptoms.
The beneficial respiratory effects of low-dose singlet oxygen stem from its ability to induce mild, transient oxidative stress, triggering a hormetic cellular response. This controlled redox perturbation activates protective signaling pathways without overwhelming endogenous antioxidant defenses.
Key mechanisms include the upregulation of antioxidant enzymes,[xix] such as superoxide dismutase, catalase, and glutathione peroxidase, modulation of pro- and anti-inflammatory mediators, and promotion of epithelial repair and regeneration.117 These responses collectively restore redox balance, attenuate tissue inflammation,[xx] reduce bronchoconstriction,[xxi] and enhance mucociliary clearance,180,181 all of which are essential for resolving respiratory infections and managing chronic airway disorders.
Additionally, singlet oxygen exposure has been proposed to stimulate autophagy and mitochondrial biogenesis, contributing to enhanced metabolic resilience[xxii] and immune competence within pulmonary tissues. These redox- and autophagy-mediated adaptations, as described in Sections 5 and 7, offer a plausible mechanistic foundation for the observed improvements in respiratory function.
Taken together, these findings support low-dose singlet oxygen as a non-invasive and physiologically adaptive modality for promoting respiratory health, particularly in conditions characterized by chronic inflammation, redox imbalance, and epithelial dysfunction.
The COVID-19 pandemic drew global attention to the devastating consequences of dysregulated immune responses in viral pneumonia, where excessive cytokine release, oxidative stress, and alveolar damage contribute to acute respiratory distress syndrome (ARDS) and long-term pulmonary complications such as fibrosis and reduced oxygen diffusion capacity.[xxiii] In the absence of curative antiviral therapies for severe cases, clinical management has relied primarily on supportive interventions such as mechanical ventilation and ECMO, both of which associated with high morbidity and long-term impairment of lung function.[xxiv]
While no controlled clinical trials have yet assessed the efficacy of low-dose SOT in COVID-19 management, a number of anecdotal reports describe apparent improvements in respiratory function, oxygenation, and symptom resolution following its use. The following case summaries are included for observational context:
Case 1: Rapid Symptom Resolution and Functional Recovery
A 15-year-old patient with confirmed COVID-19 presented with high fever (~40 °C), significant labored breathing, and loss of taste and smell. Following the initiation of SOT, the patient reported improved respiratory comfort, restoration of ambulation, and return of olfactory and gustatory function.
Case 2: Recovery from Severe Respiratory Distress
A 36-year-old recording artist experiencing severe respiratory symptoms during acute COVID-19 infection, including significant pulmonary involvement and breathing difficulties, reported progressive respiratory improvement and resumption of full functional capacity after initiating SOT.
Case 3: Prevention of Hospitalization and Respiratory Recovery
A 39-year-old woman with acute deterioration following COVID-19 infection, including loss of taste and smell, nausea, marked weakness, and episodes of respiratory distress with declining oxygen saturation, requiring emergency medical attention. After initiating SOT, she described phlegm expectoration and steady improvement in breathing and energy, with eventual discontinuation of supplemental oxygen and symptom resolution. Her family members, who also began SOT, reported similar improvement.
These reports, while anecdotal and uncontrolled, align mechanistically with the proposed redox-modulating and immunoregulatory effects of low-dose singlet oxygen described in Sections 5 and 7. Their observational consistency underscores the need for rigorous clinical trials to investigate its Physiological potential in acute viral respiratory illnesses.
Post-acute sequelae of SARS-CoV-2 infection (PASC), or long COVID, is increasingly recognized as a multifactorial syndrome characterized by persistent symptoms lasting weeks to months beyond the resolution of acute illness. According to the World Health Organization, manifestations may include fatigue, dyspnea, cognitive dysfunction (“brain fog”), autonomic instability, and multisystem involvement, often with delayed onset. [xxv]
Emerging evidence links these symptoms to chronic low-grade inflammation, endothelial dysfunction, and mitochondrial dysregulation,[xxvi] aligning closely with the redox imbalances and impaired energy metabolism described in Section 3. Long COVID pathophysiology includes microvascular injury, immune dysregulation, and altered oxygen delivery, which are all factors contributing to persistent oxidative stress and impaired cellular bioenergetics.
Low-dose singlet oxygen exposure has been anecdotally associated with improvements in these domains. Observational reports describe functional gains in respiratory endurance, cognitive clarity, and fatigue reduction, suggesting that singlet oxygen may support microcirculatory recovery, mitochondrial adaptation, and modulation of chronic inflammation.
These outcomes are consistent with the hormetic mechanisms discussed in Sections 5 and 7, particularly NRF2 activation, mitochondrial biogenesis, and autophagy induction. Although formal validation is required, the absence of systemic toxicity and the feasibility of continuous use make SOT a candidate for further research in post-viral syndromes.
While the lungs serve as an internal gateway, the skin represents the most expansive external interface for singlet oxygen-enriched air. Because the skin is naturally adapted to manage environmental oxidative stressors, such as UV radiation, it is highly sensitive to the low-dose “biophysical trigger” of singlet oxygen. Beyond its role as a physical barrier, the skin functions as an active immuno-cutaneous ecosystem where autophagy and Nrf2-mediated pathways preserve homeostatic balance. Clinical observations suggest that low-dose, ambient singlet oxygen therapy (SOT) may support the resolution of chronic inflammatory and degenerative skin conditions by activating these endogenous repair mechanisms.
Case 1: Alleviating Acne and Reducing Scarring
A female teenager described suffering from Chronic inflammatory acne, significant erythema (redness), pruritus (itchiness), and secondary psychological distress (social anxiety). Previous pharmacological interventions (topical ointments) were reported as ineffective. After utilizing SOT using a fan-like device, rapid resolution of active acne lesions and significant improvement in skin texture (smoothing of scar tissue) was observed. The young woman also reported immediate relief from acute pruritus (itchiness) associated with insect bites and emergent pimples.
Proposed Mechanism: The subject’s transition from chronic inflammation to skin clearance and scarring reduction highlights the clinical utility of the pathways established in earlier chapters:
Case 2: Relief for Atopic Dermatitis
A male infant, aged 5 months, presented with a history of severe atopic dermatitis (AD) originating at 3 weeks of age. The clinical presentation was characterized by widespread erythematous rashes and inflammatory plaques, localized primarily to the flexural folds (the inner elbows, behind the knees, neck, underarms and so on). The condition was associated with significant pruritus and cutaneous pain, resulting in chronic physical discomfort and sleep disturbances. Initial management included topical corticosteroids; however, the clinical response was transient, with symptomatic relapse occurring within 48–72 hours post-application, indicating a cycle of corticosteroid dependency and recalcitrant inflammation.
Following the introduction of nightly low-dose ambient SOT (generated via non-irradiative gas-phase technology) placed adjacent to the infant’s sleeping area, a progressive alleviation of symptoms was observed. Within six weeks of continuous nightly exposure, the inflammatory rashes achieved total clinical resolution. The cutaneous surface returned to a healthy, smooth state with no evidence of residual scaling or erythema. Long-term follow-up indicates that maintenance of nightly SOT has prevented further relapses, suggesting a stabilization of the skin barrier and a potential shift in the local inflammatory cytokine profile.
Case 3: Relief of Severe Dermatitis
A 44-year-old woman suffered for years from severe chronic eczema. Prior to treatment, the patient experienced persistent scaling and pruritus on the elbows and knees, which were only partially managed with topical corticosteroids. Within four weeks of SOT exposure, the patient observed a visible reduction in plaque thickness and redness, alongside a cessation of itching. By the eighth week, the lesions had reached near-total remission, and the patient was able to discontinue topical steroid use.
Proposed Mechanism: The observed clinical benefits in dermatological health are likely mediated by the following hormetic processes described in Sections 5 and 7:
These observations highlight SOT’s potential as a non-invasive, drug-free adjunctive therapy for adolescent acne, warranting further investigation into its effects on sebum composition and dermal cytokine profiles.
One of the earliest and most consistently reported effects by individuals undergoing SOT, including both healthy subjects and those with chronic conditions, is a subjective improvement in sleep quality. These improvements include reports of faster sleep onset, longer uninterrupted sleep, and fewer nocturnal awakenings. Such effects are notable given the central role sleep plays in physiological repair, cognitive functioning, and immune modulation.[xxviii]
Sleep is closely tied to redox biology.[xxix] During restful sleep, antioxidant systems are restored, and metabolic byproducts, including ROS, are effectively neutralized. Conversely, sleep deprivation has been shown to elevate oxidative stress in the brain and peripheral tissues, disrupt redox-sensitive signaling pathways,[xxx] and increase the production of pro-inflammatory cytokines such as TNF-α and IL-6. These inflammatory mediators can also impair sleep regulation, potentially reinforcing a feedback loop of oxidative stress, immune dysregulation,192 and poor restorative capacity[xxxi]. This may contribute to increased risk for chronic diseases such as cardiovascular and neurodegenerative disorders.[xxxii]
Case 1: Remission of Obstructive Sleep Apnea Without CPAP
A 69-year-old male diagnosed with mild-to-moderate obstructive sleep apnea (OSA) in 2018 (AHI 7.0; SpO₂ nadir 86%) was unable to tolerate continuous positive airway pressure (CPAP) therapy. Beginning in 2020–2021, he initiated nightly use of a passive, low-dose singlet oxygen-emitting device during sleep. Over the following years, he experienced a reported improvement in sleep quality and energy, coinciding with modest weight loss (BMI decreased from ~29 to 24.7).
A follow-up sleep study in 2025 revealed normalized breathing parameters (AHI 6.8, mean SpO₂ 93%, nadir 87%, sleep efficiency 91.4%), and CPAP was deemed unnecessary. Although causality cannot be established, the temporal association raises the possibility that SOT contributed to improved upper airway function and respiratory stability during sleep. This represents a rare instance of OSA remission without CPAP, surgery, or pharmacological intervention.
Proposed Mechanism: Evidence from preclinical and clinical studies suggests that moderate, transient oxidative stimuli, such as those induced by singlet oxygen, can restore redox balance through hormetic mechanisms that recalibrate circadian rhythms[xxxiii] and stress responses. This may include the resetting of dysfunctional sleep-wake cycles and attenuation of hyperarousal states commonly associated with insomnia and anxiety. [xxxiv],[xxxv]
Users commonly describe experiencing noticeable improvements within the first 24 hours of singlet oxygen exposure. These rapid effects may reflect immediate neurophysiological adaptations via redox-sensitive systems involved in melatonin synthesis, hypothalamic signaling, or neuronal hyperexcitability.[xxxvi] While speculative, the observed restoration of sleep continuity may suggest an indirect role for singlet oxygen in modulating neuroimmune interactions[xxxvii] and systemic stress resilience.
Snoring and sleep-disordered breathing are widespread issues, affecting over 40% of the population at some point in their lives with varying severity. These conditions disrupt sleep architecture by limiting airflow and preventing entry into deeper, restorative sleep phases. Snoring typically results from partial obstruction of the upper airway, often due to relaxed throat muscles, excess soft tissue, nasal congestion, or tongue positioning, leading to vibratory noise as air passes through narrowed passages.
In otherwise healthy individuals, snoring may present as a benign nuisance, typically impacting bed partners. However, in more severe presentations such as obstructive sleep apnea (OSA) or asthma, repeated airway collapse can result in intermittent hypoxia, oxidative stress, and increased cardiovascular and metabolic risks. [xxxviii]
Anecdotal reports from users of SOT have described significant reductions in snoring frequency and intensity, including among individuals not directly using the device. In over 90 reported cases, individuals receiving SOT for unrelated conditions, noted that their bed partners, many of whom were habitual snorers, experienced diminished or absent snoring during the treatment period.
One particularly striking case involved an asthma patient who began nightly SOT; shortly thereafter, his wife, previously a consistent snorer, ceased snoring entirely. This effect occurred without her direct use of the device, raising the possibility that passive exposure to singlet oxygen-enriched air may influence upper airway function.
Proposed Mechanism: Several biological mechanisms could plausibly explain these effects. Low-level oxidative stress induced by singlet oxygen may elicit hormetic responses that promote subtle increases in upper airway muscle tone,[xxxix] thereby reducing the likelihood of collapse during sleep. It may also attenuate inflammation, a key contributor to airway narrowing in conditions such as allergic rhinitis and sinus congestion. Given that OSA and snoring are associated with chronic inflammation and oxidative stress in the upper airway, the redox-balancing effects of singlet oxygen may help reduce tissue swelling, improve airflow, and lower snoring severity.
While these observations are mechanistically consistent with findings described in Sections 5 and 7, they remain preliminary. Controlled clinical studies are needed to determine the extent and reproducibility of SOT’s impact on snoring and upper airway physiology.
Chronic nightmares, that are distressing, recurrent dreams that disrupt sleep, affect approximately 2–6% of adults and are especially common in individuals with post-traumatic stress disorder (PTSD), anxiety, or depression.[xl] Despite their impact on sleep quality and mental health, nightmares are frequently underdiagnosed and undertreated. They are rarely spontaneously reported by patients and often overlooked by clinicians, despite well-documented associations with increased risk of suicide, insomnia, and emotional dysregulation.[xli],[xlii] This persistent clinical neglect has contributed to diagnostic and therapeutic gaps, leaving many individuals untreated despite significant sleep disruption and psychological distress.
Case: Resolution of Trauma-Associated Nightmares Following SOT
A woman who experienced daily trauma-related nightmares for eight years following an intraoperative awareness event reported immediate and lasting resolution of her symptoms after beginning SOT. Her symptoms, including dissociative aftereffects, reportedly ceased within the first nights of exposure, enabling sustained sleep restoration for the first time in years.
Proposed Mechanism: These effects may be mediated by redox-sensitive neural pathways involved in fear processing and REM regulation. Singlet oxygen-induced low-level oxidative stress may activate antioxidant and anti-inflammatory responses in limbic structures such as the amygdala and prefrontal cortex. These adaptations could stabilize neural activity and reduce hyperarousal, thereby improving REM sleep continuity and reducing nightmare intensity.[xliii]
Although mechanistically plausible and consistent with preclinical literature on redox-neural interactions, these observations remain anecdotal. Controlled studies are required to confirm therapeutic efficacy in nightmare disorders or trauma-related sleep disturbances.
A recent study by Hsieh et al.[xliv] reported enhanced exercise performance and physiological resilience following exposure to low-dose singlet oxygen energy (SOE). Participants using a passive, non-invasive singlet oxygen-generating device during exercise showed improvements in cardiorespiratory function, muscle recovery, and subjective vitality.
Rather than attributing these effects solely to SOE, as the writers do, they may reflect generalizable redox-based mechanisms consistent with known physiological adaptations to exercise. Physical activity induces transient oxidative stress that activates hormetic signaling pathways, including Nrf2-mediated antioxidant responses[xlv] and mitochondrial biogenesis via AMPK and PGC-1α.[xlvi] These responses are part of mitohormesis, a process by which low-level mitochondrial stress improves energy metabolism, mitochondrial quality control (via fusion/fission and mitophagy), and resistance to subsequent challenges.[xlvii]
During recovery, moderate ROS exposure stimulates AMPK signaling, facilitating glucose uptake, fatty acid oxidation, and mitochondrial renewal via upregulation of PGC-1α.[xlviii] In parallel, increased expression of antioxidant enzymes, such as superoxide dismutase and heme oxygenase-1, mitigates inflammation and protects muscle tissue from oxidative damage.[xlix] These established processes likely underlie the recovery-related benefits observed by Hsieh et al.
Thus, while the SOE device appears to confer tangible benefits, these improvements are interpretable within the broader framework of redox-based adaptive physiology, as discussed in Sections 5 and 7. Future studies comparing singlet oxygen-based delivery to other hormetic interventions, using biomarkers and controlled exercise protocols—could provide a clearer understanding of its relative efficacy.
Anemia is a condition in which there is a reduced number of red blood cells or hemoglobin molecules, which are responsible for transporting oxygen throughout the body. Consequently, reduced oxygen supply can impair tissue and organ function. In 2019, around 25% of the global population was affected by anemia (over 1.5 billion people), with the highest prevalence observed in children under five, reaching nearly 50%.[l]
One of the most common forms is anemia of chronic disease, also known as anemia of inflammation.[li] This type arises in the context of chronic illnesses such as autoimmune disorders, cancer, or kidney failure, and is characterized by impaired erythropoiesis and disturbed iron metabolism. The resulting anemia may further impair recovery by limiting oxygen delivery to tissues and suppressing immune competence.
Anecdotal reports suggest that exposure to low-level singlet oxygen may support red blood cell production, including in cases of longstanding or treatment-resistant anemia.
Case: Hematologic Improvement in a Patient with Refractory Anemia
A non-communicative adult patient with dementia, thyroid dysfunction, cardiac disease, and severe anemia (hemoglobin <8 g/dL) had not responded to conventional treatments for several years. After two months of SOT, hemoglobin levels increased to 10 g/dL and reached 10.2 g/dL after six months—levels not previously attained under standard care. Additionally, improvements in thyroid and renal function were also reported.
Proposed Mechanism: Mild oxidative stimulation may activate redox-sensitive transcription factors, supporting hematopoiesis and counteracting inflammation-induced suppression of red blood cell production.[lii] In particular, singlet oxygen may influence the hypoxia-inducible factor (HIF) pathway, which regulates erythropoietin (EPO) production in the bone marrow.[liii] Ferritin and iron metabolism may also be modulated through oxidative signaling, contributing to more effective erythropoiesis.[liv],[lv] In parallel, the mitigation of chronic inflammation via redox modulation may relieve inhibitory signals that suppress bone marrow activity in anemia of inflammation.
While these mechanisms are theoretically consistent with observed improvements, further research is essential to determine whether low-dose singlet oxygen exposure has a reproducible effect on hematologic parameters, and under which conditions it may be clinically relevant.
Chemotherapy remains one of the most commonly used and effective treatments for various types of cancer, administered in an estimated 50–60% of cases during some phase of treatment.[lvi] While its efficacy is well established, chemotherapy is frequently accompanied by debilitating side effects, including nausea, fatigue, alopecia, depression, and anemia, which can jeopardize patient compliance and survival. Reducing the severity of these adverse effects is essential to improving both treatment tolerance and outcomes.
Exposure to low levels of singlet oxygen has been anecdotally reported to reduce toxicity and improve treatment tolerance in some individuals. The following clinical observations are presented alongside plausible biological mechanisms, though they require controlled validation.
Case 1: Cancer treatment side-effects: Myelosuppression, Nausea, Fatigue, and Depression
A 50-year-old patient undergoing chemotherapy for lymphoma presented with profound exhaustion, anemia (Hb 11.2 g/dL), breathlessness, and depression. Following the initiation of SOT, the patient reportedly experienced a rapid improvement in energy and his general mood. After one month, hemoglobin levels increased to 12.6 g/dL. Subsequent rounds of chemotherapy were better tolerated, with notably reduced nausea and fatigue.
Proposed Mechanism: These effects may result from hormetic activation of protective cellular pathways, including Nrf2-mediated antioxidant responses,42 mitochondrial biogenesis,[lvii] enhanced ATP production and improved redox homeostasis.[lviii] Modulation of inflammatory signaling,[lix] and improved mitochondrial function could account for the observed improvements in fatigue and emotional resilience.
Case 2: Radiation-Induced Neuropathy and Sensory Disturbances
A patient with a history of extensive radiation therapy developed peripheral neuropathy characterized by numbness and an atypical persistent “sharp smell,” consistent with radiation-induced oxidative injury and microvascular compromise, which promote neuronal apoptosis, demyelination, and fibrotic entrapment of nerves.[lx] After initiating SOT, the patient reported a complete resolution of both sensory disturbances.
Proposed Mechanism: These outcomes may reflect activation of redox-sensitive neuroprotective pathways, including restoration of mitochondrial redox balance, Nrf2-regulated antioxidant responses, and modulation of glial-driven neuroinflammation (e.g., JNK and ERK signaling). These mechanisms are consistent with those previously discussed in Sections 5 and 7.
Dry eye disease is a chronic disorder characterized by insufficient tear production or excessive tear evaporation, resulting in ocular discomfort, surface inflammation, and visual disturbances. Its pathophysiology often involves immune-mediated processes, including macrophage activation and the release of pro-inflammatory cytokines such as interleukin-6 (IL-6) and tumor necrosis factor-alpha (TNF-α), which amplify epithelial damage and perpetuate symptoms.[lxi]
Conventional therapies, such as artificial tears, corticosteroids, and immunosuppressants like cyclosporine, primarily aim to stabilize the tear film and suppress inflammation. [lxii] More recently, antioxidant and redox-regulating strategies have emerged as promising adjuncts, given growing evidence that oxidative stress is a key contributor to dry eye pathology. [lxiii]
Anecdotal user reports suggest that exposure to low-level, non-irradiative singlet oxygen may alleviate symptoms of both chronic dry eye and allergic periorbital irritation. Users have described reduced ocular dryness and irritation, as well as rapid relief from itching, redness, and swelling commonly associated with allergic conjunctivitis. These improvements were sometimes observed within hours to days of initiating therapy.
Proposed Mechanisms
These effects may be mediated by the redox-sensitive and immune-modulatory pathways discussed earlier in the review. Singlet oxygen, when delivered at hormetic doses, likely induces mild oxidative preconditioning, which downregulates inflammatory cascades and stabilizes epithelial function. Specifically, suppression of mast cell degranulation and eosinophilic infiltration may reduce the release of histamine, leukotrienes, IL-4, IL-5, and IL-13, thereby lowering vascular permeability and sensory nerve activation.70 Additionally, SOT may restore epithelial barrier integrity through Nrf2-driven antioxidant responses (Section 5.2), increasing resistance to allergens and environmental irritants. These mechanisms may also contribute to broader immune tolerance at the mucosal surface, as previously observed in respiratory and gastrointestinal contexts (Section 7).
Taken together, these observations support the possibility that singlet oxygen may offer a non-pharmacological, mechanism-based intervention for both dry eye and allergic periorbital conditions. Rather than masking symptoms, it may address underlying biological dysfunction, illustrating a potential translational application of redox-hormetic principles.
In parallel to growing anecdotal observations presented above, academic research into singlet oxygen as a therapeutic agent is also emerging. A 2024 doctoral dissertation by Grimwood at the University of Derby[lxiv] laid the groundwork for a randomized, double-blind controlled trial investigating the physiological and psychological effects of nightly low-dose singlet oxygen exposure in individuals with COPD. While clinical results were not yet reported at the time of submission, the study involved over 180 participants and applied validated tools such as the CAT, ESS, and PSQI. Its rigorous patient-centered design and emphasis on redox-modulating therapies underscore the increasing legitimacy of this research direction and the need for peer-reviewed, controlled evaluations of clinical outcomes.
[i] Sockrider, M., & Fussner, L. (2020). What is asthma?. American journal of respiratory and critical care medicine, 202(9), P25-P26.
[ii] Venkatesan, P. (2023). 2023 GINA report for asthma. The Lancet Respiratory Medicine, 11(7), 589.
[iii] To be published
[iv] Adeloye, D., Song, P., Zhu, Y., Campbell, H., Sheikh, A., & Rudan, I. (2022). Global, regional, and national prevalence of, and risk factors for, chronic obstructive pulmonary disease (COPD) in 2019: a systematic review and modelling analysis. The Lancet Respiratory Medicine, 10(5), 447-458.
[v] Erpenbach, K. H., Brailey, M. A., & Stentiford, N. Bronchial Asthma and Chronic Obstructive Lung Diseases: Improvement of the 6. minute walking test and the lung function by inhaling activated air produced by the SOE. TIE therapeutic inhalation equipment.
[vi] Duan, K. I., Birger, M., Au, D. H., Spece, L. J., Feemster, L. C., & Dieleman, J. L. (2023). Health Care Spending on Respiratory Diseases in the United States, 1996–2016. American Journal of Respiratory and Critical Care Medicine, 207(2), 183-192.
[vii] Paulson, K. R., Kamath, A. M., Alam, T., Bienhoff, K., Abady, G. G., Abbas, J., … & Chanie, W. F. (2021). Global, regional, and national progress towards Sustainable Development Goal 3.2 for neonatal and child health: all-cause and cause-specific mortality findings from the Global Burden of Disease Study 2019. The Lancet, 398(10303), 870-905.
[viii] Gordon, B. R. (2008). Asthma history and presentation. Otolaryngologic Clinics of North America, 41(2), 375-385.
[ix] Burgel, P. R. (2012). Chronic cough and sputum production: a clinical COPD phenotype?. European Respiratory Journal, 40(1), 4-6.
[x] Diehr, P., Wood, R. W., Bushyhead, J., Krueger, L., Wolcott, B., & Tompkins, R. K. (1984). Prediction of pneumonia in outpatients with acute cough—a statistical approach. Journal of chronic diseases, 37(3), 215-225.
[xi] Tollerud, D. J., O’connor, G. T., Sparrow, D., & Weiss, S. T. (1991). Asthma, hay fever, and phlegm production associated with distinct patterns of allergy skin test reactivity, eosinophilia, and serum IgE levels. The Normative Aging Study. Am Rev Respir Dis, 144(4), 776-81.
[xii] Heijdra, Y. F., Pinto-Plata, V. M., Kenney, L. A., Rassulo, J., & Celli, B. R. (2002). Cough and phlegm are important predictors of health status in smokers without COPD. Chest, 121(5), 1427-1433.
[xiii] (a) Pitts, T., Bolser, D., Rosenbek, J., Troche, M., & Sapienza, C. (2008). Voluntary cough production and swallow dysfunction in Parkinson’s disease. Dysphagia, 23, 297-301. (b) Heijdra, Y. F., Pinto-Plata, V. M., Kenney, L. A., Rassulo, J., & Celli, B. R. (2002). Cough and phlegm are important predictors of health status in smokers without COPD. Chest, 121(5), 1427-1433. (c) Davies, D. (1974). Disability and coal workers’ pneumoconiosis. British medical journal, 2(5920), 652. (d) Warren, C. P. (1979). Acute respiratory failure and tracheal obstruction in the elderly with benign goitres. Canadian Medical Association Journal, 121(2), 191.
[xiv] Whitsett, J. A., & Alenghat, T. (2015). Respiratory epithelial cells orchestrate pulmonary innate immunity. Nature immunology, 16(1), 27-35.
[xv] Voynow, J. A., & Rubin, B. K. (2009). Mucins, mucus, and sputum. Chest, 135(2), 505-512.
[xvi] (a) Biswas, S. K., & Rahman, I. (2009). Environmental toxicity, redox signaling and lung inflammation: the role of glutathione. Molecular aspects of medicine, 30(1-2), 60-76. (b) Shao, M. X., & Nadel, J. A. (2005). Dual oxidase 1-dependent MUC5AC mucin expression in cultured human airway epithelial cells. Proceedings of the National Academy of Sciences, 102(3), 767-772.
[xvii] Price, M. E., & Sisson, J. H. (2019). Redox regulation of motile cilia in airway disease. Redox Biology, 27, 101146.
[xviii] Rancourt, R. C., Lee, R. L., O’Neill, H., Accurso, F. J., & White, C. W. (2007). Reduced thioredoxin increases proinflammatory cytokines and neutrophil influx in rat airways: modulation by airway mucus. Free Radical Biology and Medicine, 42(9), 1441-1453.
[xix] Zhang, M., Wang, J., Liu, R., Wang, Q., Qin, S., Chen, Y., & Li, W. (2024). The role of Keap1-Nrf2 signaling pathway in the treatment of respiratory diseases and the research progress on targeted drugs. Heliyon.
[xx] (a) Barnes, P. J. (2008). The cytokine network in asthma and chronic obstructive pulmonary disease. The Journal of clinical investigation, 118(11), 3546-3556. (b) Xu, W., Zhao, T., & Xiao, H. (2020). The implication of oxidative stress and AMPK-Nrf2 antioxidative signaling in pneumonia pathogenesis. Frontiers in Endocrinology, 11, 400.
[xxi] Kume, H., Yamada, R., Sato, Y., & Togawa, R. (2023). Airway smooth muscle regulated by oxidative stress in COPD. Antioxidants, 12(1), 142.
[xxii] Michaeloudes, C., Abubakar-Waziri, H., Lakhdar, R., Raby, K., Dixey, P., Adcock, I. M., … & Chung, K. F. (2022). Molecular mechanisms of oxidative stress in asthma. Molecular aspects of medicine, 85, 101026.
[xxiii] (a) Zhou, F., Yu, T., Du, R., Fan, G., Liu, Y., Liu, Z., … & Cao, B. (2020). Clinical course and risk factors for mortality of adult inpatients with COVID-19 in Wuhan, China: a retrospective cohort study. The lancet, 395(10229), 1054-1062. (b) George, P. M., Barratt, S. L., Condliffe, R., Desai, S. R., Devaraj, A., Forrest, I., … & Spencer, L. G. (2020). Respiratory follow-up of patients with COVID-19 pneumonia. Thorax, 75(11), 1009-1016.
[xxiv] Wu, Z., & McGoogan, J. M. (2020). Characteristics of and important lessons from the coronavirus disease 2019 (COVID-19) outbreak in China: summary of a report of 72 314 cases from the Chinese Center for Disease Control and Prevention. jama, 323(13), 1239-1242.
[xxv] “Post COVID-19 condition (long COVID).” World Health Organization (WHO), 26 February 2025, https://www.who.int/news-room/fact-sheets/detail/post-covid-19-condition-(long-covid). Accessed 2 August 2025.
[xxvi] (a) Nalbandian, A., Sehgal, K., Gupta, A., Madhavan, M. V., McGroder, C., Stevens, J. S., … & Wan, E. Y. (2021). Post-acute COVID-19 syndrome. Nature medicine, 27(4), 601-615. (b) Yong, S. J. (2021). Persistent brainstem dysfunction in long-COVID: a hypothesis. ACS chemical neuroscience, 12(4), 573-580.
[xxvii] Sakaguchi, R., & Mori, Y. (2020). Transient receptor potential (TRP) channels: Biosensors for redox environmental stimuli and cellular status. Free Radical Biology and Medicine, 146, 36-44.
[xxviii] Zielinski, M. R., McKenna, J. T., & McCarley, R. W. (2016). Functions and mechanisms of sleep. AIMS neuroscience, 3(1), 67.
[xxix] Peliciari-Garcia, R. A., Darley-Usmar, V., & Young, M. E. (2018). An overview of the emerging interface between cardiac metabolism, redox biology and the circadian clock. Free radical biology and medicine, 119, 75-84.
[xxx] (a) Reimund, E. (1994). The free radical flux theory of sleep. Medical hypotheses, 43(4), 231-233. (b) Villafuerte, G., Miguel-Puga, A., Murillo Rodríguez, E., Machado, S., Manjarrez, E., & Arias-Carrión, O. (2015). Sleep deprivation and oxidative stress in animal models: a systematic review. Oxidative medicine and cellular longevity, 2015(1), 234952.
[xxxi] (a) Bryndin, E., & Bryndina, I. (2020). Self healing of healthy condition at cellular level. Medical Case Reports and Reviews, 3, 1-4. (b) Adam, K., & Oswald, I. (1984). Sleep helps healing. British medical journal (Clinical research ed.), 289(6456), 1400.
[xxxii] (a) Garbarino, S., Lanteri, P., Bragazzi, N. L., Magnavita, N., & Scoditti, E. (2021). Role of sleep deprivation in immune-related disease risk and outcomes. Communications biology, 4(1), 1304. (b) Tobaldini, E., Costantino, G., Solbiati, M., Cogliati, C., Kara, T., Nobili, L., & Montano, N. (2017). Sleep, sleep deprivation, autonomic nervous system and cardiovascular diseases. Neuroscience & Biobehavioral Reviews, 74, 321-329.
[xxxiii] Wilking, M., Ndiaye, M., Mukhtar, H., & Ahmad, N. (2013). Circadian rhythm connections to oxidative stress: implications for human health. Antioxidants & redox signaling, 19(2), 192-208.
[xxxiv] Atrooz, F., & Salim, S. (2020). Sleep deprivation, oxidative stress and inflammation. Advances in protein chemistry and structural biology, 119, 309-336.
[xxxv] Patki, G., Solanki, N., Atrooz, F., Allam, F., & Salim, S. (2013). Depression, anxiety-like behavior and memory impairment are associated with increased oxidative stress and inflammation in a rat model of social stress. Brain research, 1539, 73-86.
[xxxvi] Mattson, M. P. (2008). Hormesis defined. Ageing research reviews, 7(1), 1-7.
[xxxvii] Besedovsky, L., Lange, T., & Haack, M. (2019). The sleep-immune crosstalk in health and disease. Physiological reviews.
[xxxviii] (a) Deary, V., Ellis, J. G., Wilson, J. A., Coulter, C., & Barclay, N. L. (2014). Simple snoring: not quite so simple after all?. Sleep medicine reviews, 18(6), 453-462. (b) Sarkis, L. M., Jones, A. C., Ng, A., Pantin, C., Appleton, S. L., & MacKay, S. G. (2023). Australasian Sleep Association position statement on consensus and evidence based treatment for primary snoring. Respirology, 28(2), 110-119.
[xxxix] Gonzalez-Rothi, E. J., Lee, K. Z., Dale, E. A., Reier, P. J., Mitchell, G. S., & Fuller, D. D. (2015). Intermittent hypoxia and neurorehabilitation. Journal of applied physiology, 119(12), 1455-1465.
[xl] Swart, M. L., Van Schagen, A. M., Lancee, J., & Van Den Bout, J. (2013). Prevalence of nightmare disorder in psychiatric outpatients. Psychotherapy and psychosomatics, 82(4), 267-268.
[xli] (a) Levin, R., & Nielsen, T. A. (2007). Disturbed dreaming, posttraumatic stress disorder, and affect distress: a review and neurocognitive model. Psychological bulletin, 133(3), 482. (b) Gieselmann, A., Ait Aoudia, M., Carr, M., Germain, A., Gorzka, R., Holzinger, B., … & Pietrowsky, R. (2019). Aetiology and treatment of nightmare disorder: State of the art and future perspectives. Journal of sleep research, 28(4), e12820.
[xlii] Krakow, B., & Zadra, A. (2006). Clinical management of chronic nightmares: imagery rehearsal therapy. Behavioral Sleep Medicine, 4(1), 45–70.
[xliii] Germain, A., Buysse, D. J., & Nofzinger, E. (2008). Sleep-specific mechanisms underlying posttraumatic stress disorder: integrative review and neurobiological hypotheses. Sleep medicine reviews, 12(3), 185-195.
[xliv] Hsieh, C. F., Huang, C. T., Chang, C. C., & Hung, T. P. (2025). Singlet Oxygen Energy for Enhancing Physiological Function and Athletic Performance. Bioengineering, 12(2), 118.
[xlv] Martinez-Canton, M., Galvan-Alvarez, V., Martin-Rincon, M., Calbet, J. A., & Gallego-Selles, A. (2024). Unlocking peak performance: The role of Nrf2 in enhancing exercise outcomes and training adaptation in humans. Free Radical Biology and Medicine, 224, 168-181.
[xlvi] Taskin, S., Celik, T., Demiryurek, S., Turedi, S., & Taskin, A. (2022). Effects of different-intensity exercise and creatine supplementation on mitochondrial biogenesis and redox status in mice. Iranian Journal of Basic Medical Sciences, 25(8), 1009.
[xlvii] Sun, Y., Jin, L., Qin, Y., Ouyang, Z., Zhong, J., & Zeng, Y. (2024). Harnessing mitochondrial stress for health and disease: Opportunities and challenges. Biology, 13(6), 394.
[xlviii] Viollet, B. (2017). The energy sensor AMPK: adaptations to exercise, nutritional and hormonal signals. Hormones, metabolism and the benefits of exercise, 13.
[xlix] Meng, Q., & Su, C. H. (2024). The impact of physical exercise on oxidative and nitrosative stress: balancing the benefits and risks. Antioxidants, 13(5), 573.
[l] World Health Organization. (2023). Accelerating anaemia reduction: Acomprehensive framework for action. World Health Organization.
[li] Weiss, G., Ganz, T., & Goodnough, L. T. (2019). Anemia of inflammation. Blood, The Journal of the American Society of Hematology, 133(1), 40-50.
[lii] Shaw, P., & Chattopadhyay, A. (2020). Nrf2–ARE signaling in cellular protection: Mechanism of action and the regulatory mechanisms. Journal of Cellular Physiology, 235(4), 3119-3130.
[liii] Dewhirst, M. W. (2009). Relationships between cycling hypoxia, HIF-1, angiogenesis and oxidative stress. Radiation research, 172(6), 653-665.
[liv] Wang, J., & Pantopoulos, K. (2011). Regulation of cellular iron metabolism. Biochemical Journal, 434(3), 365-381.
[lv] ORINO, K., LEHMAN, L., TSUJI, Y., AYAKI, H., TORTI, S. V., & TORTI, F. M. (2001). Ferritin and the response to oxidative stress. Biochemical Journal, 357(1), 241-247.
[lvi] Wilson, B. E., Jacob, S., Yap, M. L., Ferlay, J., Bray, F., & Barton, M. B. (2019). Estimates of global chemotherapy demands and corresponding physician workforce requirements for 2018 and 2040: a population-based study. The Lancet Oncology, 20(6), 769-780.
[lvii] Sano, M., & Fukuda, K. (2008). Activation of mitochondrial biogenesis by hormesis. Circulation research, 103(11), 1191-1193.
[lviii] Clavo, B., Rodríguez-Esparragón, F., Rodríguez-Abreu, D., Martínez-Sánchez, G., Llontop, P., Aguiar-Bujanda, D., … & Santana-Rodríguez, N. (2019). Modulation of oxidative stress by ozone therapy in the prevention and treatment of chemotherapy-induced toxicity: review and prospects. Antioxidants, 8(12), 588.
[lix] Li, N., & Karin, M. (1999). Is NF‐κB the sensor of oxidative stress?. The FASEB Journal, 13(10), 1137-1143.
[lx] (a) Pradat, P. F., & Delanian, S. (2013). Late radiation injury to peripheral nerves. Handbook of clinical neurology, 115, 743-758. (b) Pariset, E., Malkani, S., Cekanaviciute, E., & Costes, S. V. (2021). Ionizing radiation-induced risks to the central nervous system and countermeasures in cellular and rodent models. International Journal of Radiation Biology, 97(sup1), S132-S150. (c) Azzam, P., Mroueh, M., Francis, M., Abou Daher, A., & Zeidan, Y. H. (2020). Radiation-induced neuropathies in head and neck cancer: prevention and treatment modalities. ecancermedicalscience, 14, 1133.
[lxi] Yoon, K. C., Jeong, I. Y., Park, Y. G., & Yang, S. Y. (2007). Interleukin-6 and tumor necrosis factor-α levels in tears of patients with dry eye syndrome. Cornea, 26(4), 431-437.
[lxii] Pflugfelder, S. C., & de Paiva, C. S. (2017). The pathophysiology of dry eye disease: what we know and future directions for research. Ophthalmology, 124(11), S4-S13.
[lxiii] Böhm, E. W., Buonfiglio, F., Voigt, A. M., Bachmann, P., Safi, T., Pfeiffer, N., & Gericke, A. (2023). Oxidative stress in the eye and its role in the pathophysiology of ocular diseases. Redox biology, 68, 102967.
[lxiv] Grimwood, S. J. (2024). Profiling the Physiological and Psychological Effectiveness of Singlet Oxygen in the Management of Chronic Obstructive Pulmonary Disease (COPD) (Doctoral dissertation, University of Derby (United Kingdom)).
One of the major challenges in evaluating novel therapeutic interventions, particularly those involving subtle physiological modulation, is distinguishing true biological effects from placebo responses. Placebo responses are mediated through central nervous system circuits involving expectation, emotional modulation, and dopamine-linked reward pathways.[i] These mechanisms can produce transient symptom relief, but do not activate cellular repair pathways, alter redox signaling, or regenerate damaged tissue.[ii]
In the case of singlet oxygen-enriched air, several cases presented in this review provide strong evidence that the physiological benefits associated with low-dose singlet oxygen are not merely psychological. These observations, particularly in individuals who were unaware of the intervention or incapable of expectancy-based responses, provide strong evidence for a biological, rather than psychological, basis for the effects.
As previously described in the section on mucociliary function, a severely brain-injured young man, fully bedridden and non-communicative, experienced a sustained and documented reduction in the need for routine suctioning following the introduction of the singlet oxygen-enriched air generator. The cessation of routine suctioning, previously required nightly to prevent choking, occurred without any awareness or behavioral change on the part of the patient, offering compelling support for a direct physiological effect of the therapy on mucociliary function.
A similar conclusion can be drawn from the case reported under the “anemia and blood disorders” section, where an elderly man with dementia and chronic illness exhibited a sustained rise in hemoglobin levels after exposure to singlet oxygen-enriched air. Given his cognitive impairment and lack of knowledge regarding the intervention, the improvement in anemia, thyroid, and renal parameters could not have been driven by expectancy effects.
In the context of oncological support, the case of a terminal lymphoma patient, documented under supporting cancer treatment and recovery, showed an unexpectedly rapid return of verbal communication, appetite, and increased energy shortly after the initiation of low-level ambient singlet oxygen generator use. Although this patient may have been marginally aware of environmental changes, the timing, magnitude, and multifaceted nature of the improvements, including enhanced chemotherapy tolerance and rising hemoglobin levels, suggest physiological mechanisms were at play.
Further support comes from multiple reports involving young children and infants, discussed under respiratory conditions and excess phlegm, who experienced improvements in breathing, mucus clearance, and sleep quality. Given their developmental stage and limited awareness, placebo responses are not a plausible explanation in these cases.
These observations, taken together across multiple populations and physiological systems, indicate that the benefits of singlet oxygen exposure are not the result of suggestion or expectation. Instead, they reflect a pattern of redox-modulated physiological responses consistent with hormetic activation of repair and defense pathways.
[i] (a) Price, D. D., Finniss, D. G., & Benedetti, F. (2008). A comprehensive review of the placebo effect: recent advances and current thought. Annu. Rev. Psychol., 59(1), 565-590. (b) Enck, P., Benedetti, F., & Schedlowski, M. (2008). New insights into the placebo and nocebo responses. Neuron, 59(2), 195-206.
[ii] (a) Colloca, L., & Barsky, A. J. (2020). Placebo and nocebo effects. New England Journal of Medicine, 382(6), 554-561. (b) Benedetti, F., & Amanzio, M. (2013). Mechanisms of the placebo response. Pulmonary pharmacology & therapeutics, 26(5), 520-523.
Low-dose exposure to singlet oxygen represents a fundamentally different therapeutic strategy compared to conventional pharmaceuticals. While most drugs act by targeting specific receptors or pathways, singlet oxygen functions mainly through hormesis, which is a mild oxidative stimulus that activates broad, evolutionarily conserved defense systems. These include autophagy, upregulation of antioxidant enzymes, and metabolic adaptation. 106,144
Such pathways are self-limiting, highly regulated, and sensitive to context. This enables singlet oxygen to maintain physiological balance without the risks of chronic overstimulation or suppression often associated with pharmacological agents.[i] These characteristics make it especially relevant to preventive medicine, where early modulation of redox signaling can reduce inflammation, enhance cellular resilience, and delay disease progression. 124
Moreover, unlike many drug-based interventions, the protective responses triggered by singlet oxygen are not heavily influenced by individual genetic polymorphisms or metabolic profiles. This broad compatibility increases its potential applicability across diverse populations.144 Together, these features position singlet oxygen as a novel, non-pharmacological tool for modulating redox-sensitive biological networks to prevent disease onset and maintain long-term physiological equilibrium.
[i] Liu, S., Pi, J., & Zhang, Q. (2022). Signal amplification in the KEAP1-NRF2-ARE antioxidant response pathway. Redox biology, 54, 102389.
This review highlights the emerging role of singlet oxygen as a hormetic agent capable of inducing protective and adaptive cellular responses when delivered at low concentrations. Historically regarded primarily as a cytotoxic species, singlet oxygen is now recognized for its ability to modulate key signaling pathways that regulate antioxidant defenses, autophagy, and inflammation.
By activating regulatory circuits such as the Nrf2-Keap1-SIRT6 axis, MAPK signaling, and JNK-mediated autophagy, mild oxidative stress initiated by singlet oxygen promotes detoxification, cellular repair, and long-term resilience to environmental and physiological challenges. In this context, SIRT6 acts as a vital epigenetic and metabolic switch, amplifying Nrf2-dependent antioxidant transcription while simultaneously maintaining mitochondrial integrity and suppressing pro-inflammatory NF-κB signaling.
These mechanistic insights are supported by case observations presented in this review, which document improvements in a wide array of conditions, including asthma, COPD, sleep disturbances, anemia, and chemotherapy-related fatigue, following exposure to singlet oxygen-enriched air. The singlet oxygen-enriched air generator, which enables safe and non-invasive delivery of singlet oxygen under ambient conditions, represents a promising tool for translating these physiological mechanisms into therapeutic and preventive applications.
Together, the evidence supports the view that low-dose singlet oxygen can serve as a valuable adjunct in regenerative, anti-inflammatory, and resilience-enhancing interventions. The preservation and activation of central longevity proteins like SIRT6 likely underpin the systemic benefits observed, thus laying the groundwork for wider integration of singlet oxygen technology into health-supportive and anti-aging strategies.
The clinical observations and anecdotal reports presented in this review are derived from publicly accessible video and audio testimonials available online across various platforms and languages. These public testimonials have been carefully reviewed and verified by the author for relevance to the discussed topics. While these qualitative accounts are not structured datasets suitable for conventional repository deposition, their public availability enhances transparency. Further formal clinical studies are warranted to systematically validate these observations.
This article is a scientific review and does not present any new research involving human or animal participants requiring ethical approval. All anecdotal reports and observational cases mentioned are derived from publicly accessible video and audio testimonials, described in aggregate, and without any identifying information. The original consent for public dissemination of these testimonials rests with the individuals who provided them. Therefore, no additional ethical approval or individual consent was required for this review.
Dr. Yael Abraham serves as an independent consultant for the patent registration and scientific aspects related to the low-dose singlet oxygen technology (patented in the U.S., South Korea, China, India, Mexico, Europe, and other countries worldwide). She is not involved in the marketing or sales of products derived from this technology and receives no compensation from sales volume. Her current research focuses on the biological effects of low-dose singlet oxygen. This consulting role does not influence the content or conclusions of this review.
The author declared that no grants were involved in supporting this work
Not applicable.
The author was responsible for scientific consulting during the development of the device, gathering clinical observations from users, and interpreting findings through existing biomedical literature. The author also prepared and wrote the review manuscript.
The author acknowledges the use of OpenAI’s ChatGPT (version GPT-4, June 2025) in the preparation of this manuscript. ChatGPT was used under the guidance of the lead author to assist with tasks such as grammar editing, scientific rephrasing, summarizing peer-reviewed literature, and structuring scientific content. All content generated was critically reviewed, validated against original sources, and edited by the author to ensure scientific accuracy and integrity.
[1] (a) Klotz, L. O., Kröncke, K. D., & Sies, H. (2003). Singlet oxygen-induced signaling effects in mammalian cells. Photochemical & photobiological sciences, 2(2), 88-94. (b) Jones, D. P., & Sies, H. (2015). The redox code. Antioxidants & redox signaling, 23(9), 734-746.
[1] Zheng, C., Chen, J. P., Wang, X. W., & Li, P. (2025). Reactive Oxygen Species in Plants: Metabolism, Signaling, and Oxidative Modifications. Antioxidants, 14(6), 617.
[1] Murotomi, K., Umeno, A., Shichiri, M., Tanito, M., & Yoshida, Y. (2023). Significance of singlet oxygen molecule in pathologies. International Journal of Molecular Sciences, 24(3), 2739.
[1] Calabrese, E. J., & Mattson, M. P. (2011). Hormesis provides a generalized quantitative estimate of biological plasticity. Journal of cell communication and signaling, 5(1), 25-38.
[1] Wilkinson, F., Helman, W. P., & Ross, A. B. (1995). Rate constants for the decay and reactions of the lowest electronically excited singlet state of molecular oxygen in solution. An expanded and revised compilation. Journal of Physical and Chemical Reference Data, 24(2), 663-677.
[1] (a) Hasegawa, K., Yamada, K., Sasase, R., Miyazaki, R., Kikuchi, A., & Yagi, M. (2008). Direct measurements of absolute concentration and lifetime of singlet oxygen in the gas phase by electron paramagnetic resonance. Chemical Physics Letters, 457(4-6), 312-314. (b) Wang, K. K., Song, S., Jung, S. J., Hwang, J. W., Kim, M. G., Kim, J. H., … & Kim, Y. R. (2020). Lifetime and diffusion distance of singlet oxygen in air under everyday atmospheric conditions. Physical Chemistry Chemical Physics, 22(38), 21664-21671.
[1] Hurst, J. R., McDonald, J. D., & Schuster, G. B. (1982). Lifetime of singlet oxygen in solution directly determined by laser spectroscopy. Journal of the American Chemical Society, 104(7), 2065-2067.
[1] (a) Noronha-Dutra, A. A., Epperlein, M. M., & Woolf, N. (1993). Reaction of nitric oxide with hydrogen peroxide to produce potentially cytotoxic singlet oxygen as a model for nitric oxide-mediated killing. FEBS letters, 321(1), 59-62. (b) Kanofsky, J. R. (1984). Singlet oxygen production by chloroperoxidase-hydrogen peroxide-halide systems. Journal of Biological Chemistry, 259(9), 5596-5600. (c) Foote, C. S., Wexler, S., Ando, W., & Higgins, R. (1968). Chemistry of singlet oxygen. IV. Oxygenations with hypochlorite-hydrogen peroxide. Journal of the American Chemical Society, 90(4), 975-981. (d) Stephenson, L. M., & McClure, D. E. (1973). Mechanisms in phosphite ozonide decomposition to phosphate esters and singlet oxygen. Journal of the American Chemical Society, 95(9), 3074-3076.
[1] Barry Halliwell; John MC. (1982) Free Radical in Biology and Medicine. Second Edition. Clarwndon Press. OxFord.
[1] Makuch, S., Dróżdż, M., Makarec, A., Ziółkowski, P., & Woźniak, M. (2022). An Update on Photodynamic Therapy of Psoriasis—Current Strategies and Nanotechnology as a Future Perspective. International journal of molecular sciences, 23(17), 9845.
[1] Jiang, J., Lv, X., Cheng, H., Yang, D., Xu, W., Hu, Y., … & Zeng, G. (2024). Type I photodynamic antimicrobial therapy: principles, progress, and future perspectives. Acta Biomaterialia, 177, 1-19.
[1] Singh, S., & Awasthi, R. (2023). Breakthroughs and bottlenecks of psoriasis therapy: Emerging trends and advances in lipid based nano-drug delivery platforms for dermal and transdermal drug delivery. Journal of Drug Delivery Science and Technology, 104548.
[1] DeRosa, M. C., & Crutchley, R. J. (2002). Photosensitized singlet oxygen and its applications. Coordination Chemistry Reviews, 233, 351-371.
[1] (a) Eisenberg, W. C., & DeSilva, M. (1990). Atmospheric gas phase generation of singlet oxygen by homogeneous photosensitization. Tetrahedron letters, 31(41), 5857-5860. (b) Eisenberg, W. C., & DeSilva, M. (1990). Atmospheric gas phase generation of singlet oxygen by homogeneous photosensitization. Tetrahedron letters, 31(41), 5857-5860. (c) Funken, K. H., Horneck, G., Milow, B., Schafer, M., Schmitz, C., Faust, D., … & Sattlegger, M. (2000). U.S. Patent No. 6,107,480. Washington, DC: U.S. Patent and Trademark Office. (d) Sunday, M. O., & Sakugawa, H. (2020). A simple, inexpensive method for gas-phase singlet oxygen generation from sensitizer-impregnated filters: Potential application to bacteria/virus inactivation and pollutant degradation. Science of the Total Environment, 746, 141186.
[1] (1) Ihalagedara, H. B., Xu, Q., Greer, A., & Lyons, A. M. (2025). Singlet oxygen generation on a superhydrophobic surface: Effect of photosensitizer coating and incident wavelength on 1O2 yields. Photochemistry and Photobiology, 101(1), 167-179. (2) Arpa, E. M., & Corral, I. (2023). Unveiling Photodegradation and Photosensitization Mechanisms of Unconjugated Pterins. Chemistry–A European Journal, 29(29), e202300519.
[1] (a) Gorbanev, Y., & Bogaerts, A. (2018). Chemical detection of short-lived species induced in aqueous media by atmospheric pressure plasma. In Atmospheric Pressure Plasma-from Diagnostics to Applications. IntechOpen. (b) Cabrellon, G., Tampieri, F., Rossa, A., Barbon, A., Marotta, E., & Paradisi, C. (2020). Application of fluorescence-based probes for the determination of superoxide in water treated with air non-thermal plasma. ACS sensors, 5(9), 2866-2875. (c) Aggelopoulos, C. A., Tataraki, D., & Rassias, G. (2018). Degradation of atrazine in soil by dielectric barrier discharge plasma–potential singlet oxygen mediation. Chemical Engineering Journal, 347, 682-694. (d) Jablonowski, H., Santos Sousa, J., Weltmann, K. D., Wende, K., & Reuter, S. (2018). Quantification of the ozone and singlet delta oxygen produced in gas and liquid phases by a non-thermal atmospheric plasma with relevance for medical treatment. Scientific reports, 8(1), 12195. (e) Lim, J., Park, S., Ryu, S., Park, S., & Kim, M. S. (2025). Different inactivation mechanisms of Staphylococcus aureus and Escherichia coli in water by reactive oxygen and nitrogen species generated from an argon plasma jet. Environmental Science & Technology, 59(6), 3276-3285.
[1] (a) Kuk, S. K., Ji, S. M., Kang, S., Yang, D. S., Kwon, H. J., Koo, M. S., … & Lee, H. C. (2023). Singlet-oxygen-driven photocatalytic degradation of gaseous formaldehyde and its mechanistic study. Applied Catalysis B: Environmental, 328, 122463. (b) Zollo, A., Livraghi, S., Giamello, E., Cioni, A., Dami, V., Lorenzi, G., … & Zaleska-Medynska, A. (2023). How to guide photocatalytic applications of titanium dioxide co-doped with nitrogen and carbon by modulating the production of reactive oxygen species. Journal of Environmental Chemical Engineering, 11(6), 111523. (c) Li, Q., Zhao, J., Shang, H., Ma, Z., Cao, H., Zhou, Y., … & Li, H. (2022). Singlet Oxygen and Mobile Hydroxyl Radicals Co-operating on Gas–solid Catalytic Reaction Interfaces for Deeply Oxidizing NO x. Environmental Science & Technology, 56(9), 5830-5839. (d) Ibhadon, A. O., & Fitzpatrick, P. (2013). Heterogeneous photocatalysis: recent advances and applications. Catalysts, 3(1), 189-218. (e) Shi, Y., Yang, Z., Shi, L., Li, H., Liu, X., Zhang, X., … & Zhang, L. (2022). Surface boronizing can weaken the excitonic effects of BiOBr nanosheets for efficient O2 activation and selective NO oxidation under visible light irradiation. Environmental Science & Technology, 56(20), 14478-14486. (f) Yaghmaei Sabegh, M. (2024). Advancements in Photochemistry and Flow Systems for Synthesis and Analysis (Doctoral dissertation, Université d’Ottawa| University of Ottawa).
[1] Badash, Z. (2021). U.S. Patent No. 11,007,129. Washington, DC: U.S. Patent and Trademark Office.
[1] Carbogno, C., Groß, A., Meyer, J., & Reuter, K. (2013). O2 Adsorption Dynamics at Metal Surfaces: Non-Adiabatic Effects, Dissociation and Dissipation. In Dynamics of Gas-Surface Interactions (pp. 389-419). Springer Berlin Heidelberg.
[1] Devasagayam, T., & Kamat, J. P. (2002). Biological significance of singlet oxygen.
[1] Dmitrieva, V. A., Tyutereva, E. V., & Voitsekhovskaja, O. V. (2020). Singlet oxygen in plants: Generation, detection, and signaling roles. International journal of molecular sciences, 21(9), 3237.
[1] Kiryu, C., Makiuchi, M., Miyazaki, J., Fujinaga, T., & Kakinuma, K. (1999). Physiological production of singlet molecular oxygen in the myeloperoxidase-H2O2-chloride system. FEBS letters, 443(2), 154-158.
[1] Sun, Y., Lu, Y., Saredy, J., Wang, X., Drummer IV, C., Shao, Y., … & Yang, X. (2020). ROS systems are a new integrated network for sensing homeostasis and alarming stresses in organelle metabolic processes. Redox biology, 37, 101696.
[1] Sokolovski, S. G., Rafailov, E. U., Abramov, A. Y., & Angelova, P. R. (2021). Singlet oxygen stimulates mitochondrial bioenergetics in brain cells. Free Radical Biology and Medicine, 163, 306-313.
[1] Szechyńska-Hebda, M., Ghalami, R. Z., Kamran, M., Van Breusegem, F., & Karpiński, S. (2022). To Be or Not to Be? Are Reactive Oxygen Species, Antioxidants, and Stress Signalling Universal Determinants of Life or Death? Cells, 11(24), 4105.
[1] Davies, M. J. (2004). Reactive species formed on proteins exposed to singlet oxygen. Photochemical & Photobiological Sciences, 3, 17-25.
[1] (a) Di Mascio, P., Martinez, G. R., Miyamoto, S., Ronsein, G. E., Medeiros, M. H., & Cadet, J. (2019). Singlet molecular oxygen reactions with nucleic acids, lipids, and proteins. Chemical reviews, 119(3), 2043-2086. (b) Juan, C. A., Pérez de la Lastra, J. M., Plou, F. J., & Pérez-Lebeña, E. (2021). The chemistry of reactive oxygen species (ROS) revisited: outlining their role in biological macromolecules (DNA, lipids and proteins) and induced pathologies. International Journal of Molecular Sciences, 22(9), 4642.
[1] (a) Forman, H. J., & Zhang, H. (2021). Targeting oxidative stress in disease: promise and limitations of antioxidant therapy. Nature Reviews Drug Discovery, 20(9), 689-709. (b) Brieger, K., Schiavone, S., Miller Jr, F. J., & Krause, K. H. (2012). Reactive oxygen species: from health to disease. Swiss medical weekly, 142, w13659. (c) Rahman, T., Hosen, I., Islam, M. T., & Shekhar, H. U. (2012). Oxidative stress and human health. Advances in Bioscience and Biotechnology, 3(07), 997.
[1] Dmitrieva, V. A., Tyutereva, E. V., & Voitsekhovskaja, O. V. (2020). Singlet Oxygen in Plants: Generation, Detection, and Signaling Roles. International journal of molecular sciences, 21(9), 3237.
[1] Saed-Moucheshi, A., Sohrabi, F., & Shirkhani, A. (2023). A review on reactive oxygen species (ROS): production, function, and their influence on plants. Crop Biotechnology, 13(44), 53-70.
[1] Ziegelhoffer, E. C., & Donohue, T. J. (2009). Bacterial responses to photo-oxidative stress. Nature Reviews Microbiology, 7(12), 856-863.
[1] Maharjan, P. S., & Bhattarai, H. K. (2022). Singlet oxygen, photodynamic therapy, and mechanisms of cancer cell death. Journal of oncology, 2022(1), 7211485.
[1] Poli, G., Leonarduzzi, G., Biasi, F., & Chiarpotto, E. (2004). Oxidative stress and cell signalling. Current medicinal chemistry, 11(9), 1163-1182.
[1] (a) Iuliano, L. (2011). Pathways of cholesterol oxidation via non-enzymatic mechanisms. Chemistry and physics of lipids, 164(6), 457-468. (b) Kulig, W., Olżyńska, A., Jurkiewicz, P., Kantola, A. M., Komulainen, S., Manna, M., … & Jungwirth, P. (2015). Cholesterol under oxidative stress—How lipid membranes sense oxidation as cholesterol is being replaced by oxysterols. Free radical biology and medicine, 84, 30-41.
[1] (a) Lorenzen, I., Eble, J. A., & Hanschmann, E. M. (2021). Thiol switches in membrane proteins-Extracellular redox regulation in cell biology. Biological chemistry, 402(3), 253-269. (b) Bigelow, D. J., & Squier, T. C. (2011). Thioredoxin-dependent redox regulation of cellular signaling and stress response through reversible oxidation of methionines. Molecular Biosystems, 7(7), 2101-2109.
[1] Gems, D., & Partridge, L. (2008). Stress-response hormesis and aging:“that which does not kill us makes us stronger”. Cell metabolism, 7(3), 200-203.
[1] Shaw, P., & Chattopadhyay, A. (2020). Nrf2–ARE signaling in cellular protection: Mechanism of action and the regulatory mechanisms. Journal of Cellular Physiology, 235(4), 3119-3130.
[1] Baird, L., Llères, D., Swift, S., & Dinkova-Kostova, A. T. (2013). Regulatory flexibility in the Nrf2-mediated stress response is conferred by conformational cycling of the Keap1-Nrf2 protein complex. Proceedings of the National Academy of Sciences, 110(38), 15259-15264.
[1] Liu, X., Ren, S., Li, Z., Hao, D., Zhao, X., Zhang, Z., & Liu, D. (2023). Sirt6 mediates antioxidative functions by increasing Nrf2 abundance. Experimental cell research, 422(1), 113409.
[1] Rezazadeh, S., Yang, D., Tombline, G., Simon, M., Regan, S. P., Seluanov, A., & Gorbunova, V. (2019). SIRT6 promotes transcription of a subset of NRF2 targets by mono-ADP-ribosylating BAF170. Nucleic acids research, 47(15), 7914-7928.
[1] Lu, S. C. (2013). Glutathione synthesis. Biochimica et Biophysica Acta (BBA)-General Subjects, 1830(5), 3143-3153.
[1] Kensler, T. W., Wakabayashi, N., & Biswal, S. (2007). Cell survival responses to environmental stresses via the Keap1-Nrf2-ARE pathway. Annu. Rev. Pharmacol. Toxicol., 47(1), 89-116.
[1] Conrad, M., & Pratt, D. A. (2019). The chemical basis of ferroptosis. Nature chemical biology, 15(12), 1137-1147.
[1] Flohé, L., Toppo, S., & Orian, L. (2022). The glutathione peroxidase family: Discoveries and mechanism. Free Radical Biology and Medicine, 187, 113-122.
[1] Ma, Q. (2013). Role of nrf2 in oxidative stress and toxicity. Annual review of pharmacology and toxicology, 53(1), 401-426.
[1] Grek, C. L., Zhang, J., Manevich, Y., Townsend, D. M., & Tew, K. D. (2013). Causes and consequences of cysteine S-glutathionylation. Journal of Biological Chemistry, 288(37), 26497-26504.
[1] Telorack, M., Meyer, M., Ingold, I., Conrad, M., Bloch, W., & Werner, S. (2016). A glutathione-Nrf2-thioredoxin cross-talk ensures keratinocyte survival and efficient wound repair. PLoS genetics, 12(1), e1005800.
[1] (a) Ross, D., & Siegel, D. (2021). The diverse functionality of NQO1 and its roles in redox control. Redox Biology, 41, 101950. (b) Lee, W. S., Ham, W., & Kim, J. (2021). Roles of NAD (P) H: quinone oxidoreductase 1 in diverse diseases. Life, 11(12), 1301.
[1] (a) Waza, A. A., Hamid, Z., Ali, S., Bhat, S. A., & Bhat, M. A. (2018). A review on heme oxygenase-1 induction: is it a necessary evil. Inflammation Research, 67, 579-588. (b) Loboda, A., Damulewicz, M., Pyza, E., Jozkowicz, A., & Dulak, J. (2016). Role of Nrf2/HO-1 system in development, oxidative stress response and diseases: an evolutionarily conserved mechanism. Cellular and molecular life sciences, 73, 3221-3247. (c) Son, Y., Lee, J. H., Chung, H. T., & Pae, H. O. (2013). Therapeutic roles of heme oxygenase‐1 in metabolic diseases: curcumin and resveratrol analogues as possible inducers of heme oxygenase‐1. Oxidative medicine and cellular longevity, 2013(1), 639541.
[1] Knoops, B., Argyropoulou, V., Becker, S., Ferté, L., & Kuznetsova, O. (2016). Multiple roles of peroxiredoxins in inflammation. Molecules and cells, 39(1), 60-64.
[1] Bafana, A., Dutt, S., Kumar, A., Kumar, S., & Ahuja, P. S. (2011). The basic and applied aspects of superoxide dismutase. Journal of Molecular Catalysis B: Enzymatic, 68(2), 129-138.
[1] Kirkman, H. N., & Gaetani, G. F. (2007). Mammalian catalase: a venerable enzyme with new mysteries. Trends in biochemical sciences, 32(1), 44-50.
[1] Lu, J., & Holmgren, A. (2014). The thioredoxin antioxidant system. Free radical biology and medicine, 66, 75-87.
[1] Greeshma, M. V., Baidya, A., Mabalirajan, U., Madhunapantula, S. V., Thimmulappa, R. K., & Mahesh, P. A. (2024). Deciphering the role of 12/15-lipoxygenase in asthma: insights into mitochondrial dysfunction and therapeutic implications. Exploration of Asthma & Allergy, 2(6), 529-550.
[1] Zhang, J., Zhang, M., Tatar, M., & Gong, R. (2025). Keap1-independent Nrf2 regulation: A novel therapeutic target for treating kidney disease. Redox Biology, 103593.
[1] Dong, H., Lyu, Y., Huang, C. Y., & Tsai, S. Y. (2025). Limiting cap-dependent translation increases 20S proteasomal degradation and protects the proteomic integrity in autophagy-deficient skeletal muscle. Autophagy, 21(6), 1212-1227.
[1] Kiang, J. G., & Tsokos, G. C. (1998). Heat shock protein 70 kDa: molecular biology, biochemistry, and physiology. Pharmacology & therapeutics, 80(2), 183-201.
[1] Ameri, K., & Harris, A. L. (2008). Activating transcription factor 4. The international journal of biochemistry & cell biology, 40(1), 14-21.
[1] (a) Richter-Landsberg, C., Wyttenbach, A., & Arrigo, A. P. (2009). The role of heat shock proteins during neurodegeneration in Alzheimer’s, Parkinson’s and Huntington’s disease. Heat shock proteins in neural cells, 81-99. (b) Zhang, N., Nao, J., Zhang, S., & Dong, X. (2024). Novel insights into the activating transcription factor 4 in Alzheimer’s disease and associated aging-related diseases: Mechanisms and therapeutic implications. Frontiers in Neuroendocrinology, 101144.
[1] (a) Bjørklund, G., Zou, L., Peana, M., Chasapis, C. T., Hangan, T., Lu, J., & Maes, M. (2022). The role of the thioredoxin system in brain diseases. Antioxidants, 11(11), 2161. (b) Cimini, A., Gentile, R., Angelucci, F., Benedetti, E., Pitari, G., Giordano, A., & Ippoliti, R. (2013). Neuroprotective effects of PrxI over‐expression in an in vitro human Alzheimer’s disease model. Journal of cellular biochemistry, 114(3), 708-715. (c) Zeng, X. S., Jia, J. J., Kwon, Y., Wang, S. D., & Bai, J. (2014). The role of thioredoxin-1 in suppression of endoplasmic reticulum stress in Parkinson disease. Free Radical Biology and Medicine, 67, 10-18.
[1] (a) Ahsan, M. K., Lekli, I., Ray, D., Yodoi, J., & Das, D. K. (2009). Redox regulation of cell survival by the thioredoxin superfamily: an implication of redox gene therapy in the heart. Antioxidants & redox signaling, 11(11), 2741-2758. (b) Chong, C. R., Chan, W. P. A., Nguyen, T. H., Liu, S., Procter, N. E., Ngo, D. T., … & Horowitz, J. D. (2014). Thioredoxin-interacting protein: pathophysiology and emerging pharmacotherapeutics in cardiovascular disease and diabetes. Cardiovascular drugs and therapy, 28, 347-360.
[1] Hao, X., Zhao, B., Towers, M., Liao, L., Monteiro, E. L., Xu, X., … & Zhang, R. (2024). TXNRD1 drives the innate immune response in senescent cells with implications for age-associated inflammation. Nature Aging, 4(2), 185-197.
[1] Yang, Z. F., Drumea, K., Mott, S., Wang, J., & Rosmarin, A. G. (2014). GABP transcription factor (nuclear respiratory factor 2) is required for mitochondrial biogenesis. Molecular and cellular biology, 34(17), 3194-3201.
[1] (a) Gureev, A. P., Shaforostova, E. A., & Popov, V. N. (2019). Regulation of mitochondrial biogenesis as a way for active longevity: interaction between the Nrf2 and PGC-1α signaling pathways. Frontiers in genetics, 10, 435. (b) St-Pierre, J., Drori, S., Uldry, M., Silvaggi, J. M., Rhee, J., Jäger, S., … & Spiegelman, B. M. (2006). Suppression of reactive oxygen species and neurodegeneration by the PGC-1 transcriptional coactivators. Cell, 127(2), 397-408.
[1] Clements, C. M., McNally, R. S., Conti, B. J., Mak, T. W., & Ting, J. P. Y. (2006). DJ-1, a cancer-and Parkinson’s disease-associated protein, stabilizes the antioxidant transcriptional master regulator Nrf2. Proceedings of the National Academy of Sciences, 103(41), 15091-15096.
[1] (a) Maiorino, M., Conrad, M., & Ursini, F. (2018). GPx4, lipid peroxidation, and cell death: discoveries, rediscoveries, and open issues. Antioxidants & redox signaling, 29(1), 61-74. (b) Lu, L., Oveson, B. C., Jo, Y. J., Lauer, T. W., Usui, S., Komeima, K., … & Campochiaro, P. A. (2009). Increased expression of glutathione peroxidase 4 strongly protects retina from oxidative damage. Antioxidants & redox signaling, 11(4), 715-724.
[1] Roichman, A., Elhanati, S., Aon, M. A., Abramovich, I., Di Francesco, A., Shahar, Y., … & Cohen, H. Y. (2021). Restoration of energy homeostasis by SIRT6 extends healthy lifespan. Nature communications, 12(1), 3208.
[1] Cheng, J., Keuthan, C. J., & Esumi, N. (2023). The many faces of SIRT6 in the retina and retinal pigment epithelium. Frontiers in Cell and Developmental Biology, 11, 1244765.
[1] Palmeira, C. M., Teodoro, J. S., Amorim, J. A., Steegborn, C., Sinclair, D. A., & Rolo, A. P. (2019). Mitohormesis and metabolic health: The interplay between ROS, cAMP and sirtuins. Free Radical Biology and Medicine, 141, 483-491.
[1] Kobayashi, E. H., Suzuki, T., Funayama, R., Nagashima, T., Hayashi, M., Sekine, H., … & Yamamoto, M. (2016). Nrf2 suppresses macrophage inflammatory response by blocking proinflammatory cytokine transcription. Nature communications, 7(1), 1-14.
[1] Guo, Z., Li, P., Ge, J., & Li, H. (2022). SIRT6 in aging, metabolism, inflammation and cardiovascular diseases. Aging and disease, 13(6), 1787.
[1] He, Y., Yang, G., Sun, L., Gao, H., Yao, F., Jin, Z., … & Lin, R. (2021). SIRT6 inhibits inflammatory response through regulation of NRF2 in vascular endothelial cells. International immunopharmacology, 99, 107926.
[1] Xu, W. D., Yang, C., & Huang, A. F. (2024). The role of Nrf2 in immune cells and inflammatory autoimmune diseases: a comprehensive review. Expert Opinion on Therapeutic Targets, 28(9), 789-806.
[1] (a) Dodson, M., Shakya, A., Anandhan, A., Chen, J., Garcia, J. G., & Zhang, D. D. (2022). NRF2 and diabetes: the good, the bad, and the complex. Diabetes, 71(12), 2463-2476. (b) Vasileva, L. V., Savova, M. S., Amirova, K. M., Dinkova-Kostova, A. T., & Georgiev, M. I. (2020). Obesity and NRF2-mediated cytoprotection: Where is the missing link?. Pharmacological research, 156, 104760. (c) Li, S., Eguchi, N., Lau, H., & Ichii, H. (2020). The role of the Nrf2 signaling in obesity and insulin resistance. International journal of molecular sciences, 21(18), 6973.
[1] (a) Zhang, Y., Wang, J., Wang, Y., & Lei, K. (2024). Nrf2/HO-1 signaling activation alleviates cigarette smoke-induced inflammation in chronic obstructive pulmonary disease by suppressing NLRP3-mediated pyroptosis. Journal of Cardiothoracic Surgery, 19(1), 58. (b) Audousset, C., McGovern, T., & Martin, J. G. (2021). Role of Nrf2 in disease: novel molecular mechanisms and therapeutic approaches–pulmonary disease/asthma. Frontiers in Physiology, 12, 727806.
[1] Saha, S., Buttari, B., Profumo, E., Tucci, P., & Saso, L. (2022). A perspective on Nrf2 signaling pathway for neuroinflammation: a potential therapeutic target in Alzheimer’s and Parkinson’s diseases. Frontiers in cellular neuroscience, 15, 787258.
[1] Shi, T., & Dansen, T. B. (2020). Reactive oxygen species induced p53 activation: DNA damage, redox signaling, or both?. Antioxidants & Redox Signaling, 33(12), 839-859.
[1] Klotz, L. O., Briviba, K., & Sies, H. (1997). Singlet oxygen mediates the activation of JNK by UVA radiation in human skin fibroblasts. FEBS letters, 408(3), 289-291.
[1] Makino, T., Jinnin, M., Muchemwa, F. C., Fukushima, S., Kogushi‐Nishi, H., Moriya, C., … & Ihn, H. (2010). Basic fibroblast growth factor stimulates the proliferation of human dermal fibroblasts via the ERK1/2 and JNK pathways. British Journal of Dermatology, 162(4), 717-723.
[1] (a) Cuenda, A., & Rousseau, S. (2007). p38 MAP-kinases pathway regulation, function and role in human diseases. Biochimica et Biophysica Acta (BBA)-Molecular Cell Research, 1773(8), 1358-1375.
[1] Klionsky, D. J. (2005). The molecular machinery of autophagy: unanswered questions. Journal of cell science, 118(Pt 1), 7. (b) Martindale, J. L., & Holbrook, N. J. (2002). Cellular response to oxidative stress: signaling for suicide and survival. Journal of cellular physiology, 192(1), 1-15.
[1] (a) Wei, Y., Pattingre, S., Sinha, S., Bassik, M., & Levine, B. (2008). JNK1-mediated phosphorylation of Bcl-2 regulates starvation-induced autophagy. Molecular cell, 30(6), 678-688. (b) Yu, C., Minemoto, Y., Zhang, J., Liu, J., Tang, F., Bui, T. N., … & Lin, A. (2004). JNK suppresses apoptosis via phosphorylation of the proapoptotic Bcl-2 family protein BAD. Molecular cell, 13(3), 329-340.
[1] (a) Zhou, J., Li, X. Y., Liu, Y. J., Feng, J., Wu, Y., Shen, H. M., & Lu, G. D. (2022). Full-coverage regulations of autophagy by ROS: from induction to maturation. Autophagy, 18(6), 1240-1255. (b) Przygoda, M., Bartusik-Aebisher, D., Dynarowicz, K., Cieślar, G., Kawczyk-Krupka, A., & Aebisher, D. (2023). Cellular mechanisms of singlet oxygen in photodynamic therapy. International Journal of Molecular Sciences, 24(23), 16890. (c) Liang, P., Kolodieznyi, D., Creeger, Y., Ballou, B., & Bruchez, M. P. (2020). Subcellular singlet oxygen and cell death: location matters. Frontiers in chemistry, 8, 592941.
[1] (a) Davies, K. J. (2000). Oxidative stress, antioxidant defenses, and damage removal, repair, and replacement systems. IUBMB life, 50(4‐5), 279-289. (b) Gao, Q. (2019). Oxidative stress and autophagy. Autophagy: biology and diseases: basic science, 179-198.
[1] (a) Kaminskyy, V. O., & Zhivotovsky, B. (2014). Free radicals in cross talk between autophagy and apoptosis. Antioxidants & redox signaling, 21(1), 86-102. (b) Yue, J., & López, J. M. (2020). Understanding MAPK signaling pathways in apoptosis. International journal of molecular sciences, 21(7), 2346.
[1] Kang, R., Zeh, H. J., Lotze, M. T., & Tang, D. J. C. D. (2011). The Beclin 1 network regulates autophagy and apoptosis. Cell Death & Differentiation, 18(4), 571-580.
[1] Asgari, R., Yarani, R., Mohammadi, P., & Emami Aleagha, M. S. (2022). HIF-1α in the crosstalk between reactive oxygen species and autophagy process: a review in multiple sclerosis. Cellular and molecular neurobiology, 42(7), 2121-2129.
[1] Romero, N. R., & Agostinis, P. (2014). Molecular mechanisms underlying the activation of autophagy pathways by reactive oxygen species and their relevance in cancer progression and therapy. In Autophagy: Cancer, Other Pathologies, Inflammation, Immunity, Infection, and Aging (pp. 159-178). Academic Press.
[1] Horan, M. P., Pichaud, N., & Ballard, J. W. O. (2012). Quantifying mitochondrial dysfunction in complex diseases of aging. Journals of Gerontology Series A: Biomedical Sciences and Medical Sciences, 67(10), 1022-1035.
[1] Osellame, L. D., Rahim, A. A., Hargreaves, I. P., Gegg, M. E., Richard-Londt, A., Brandner, S., … & Duchen, M. R. (2013). Mitochondria and quality control defects in a mouse model of Gaucher disease—links to Parkinson’s disease. Cell metabolism, 17(6), 941-953.
[1] Morciano, G., Patergnani, S., Bonora, M., Pedriali, G., Tarocco, A., Bouhamida, E., … & Pinton, P. (2020). Mitophagy in cardiovascular diseases. Journal of clinical medicine, 9(3), 892.
[1] Miao, M. Q., Han, Y. B., & Liu, L. (2023). Mitophagy in metabolic syndrome. The Journal of Clinical Hypertension, 25(5), 397-403.
[1] Park, J. H., Ko, J., Park, Y. S., Park, J., Hwang, J., & Koh, H. C. (2017). Clearance of damaged mitochondria through PINK1 stabilization by JNK and ERK MAPK signaling in chlorpyrifos-treated neuroblastoma cells. Molecular neurobiology, 54, 1844-1857.
[1] (a) Chen, M., Chen, Z., Wang, Y., Tan, Z., Zhu, C., Li, Y., … & Chen, Q. (2016). Mitophagy receptor FUNDC1 regulates mitochondrial dynamics and mitophagy. Autophagy, 12(4), 689-702. (b) Park, S. Y., & Koh, H. C. (2020). FUNDC1 regulates receptor-mediated mitophagy independently of the PINK1/Parkin-dependent pathway in rotenone-treated SH-SY5Y cells. Food and Chemical Toxicology, 137, 111163. (c) He, H., Huang, W., Xiong, L., Ma, C., Wang, Y., Sun, P., … & Wu, Y. (2024). FUNDC1-mediated mitophagy regulates photodamage independently of the PINK1/Parkin-dependent pathway. Free Radical Biology and Medicine, 225, 630-640.
[1] Majeski, A. E., & Dice, J. F. (2004). Mechanisms of chaperone-mediated autophagy. The international journal of biochemistry & cell biology, 36(12), 2435-2444.
[1] Xiong, Y., Contento, A. L., Nguyen, P. Q., & Bassham, D. C. (2007). Degradation of oxidized proteins by autophagy during oxidative stress in Arabidopsis. Plant physiology, 143(1), 291-299.
[1] Kiffin, R., Christian, C., Knecht, E., & Cuervo, A. M. (2004). Activation of chaperone-mediated autophagy during oxidative stress. Molecular biology of the cell, 15(11), 4829-4840.
[1] Cuervo, A. M., & Dice, J. F. (2000). Unique properties of lamp2a compared to other lamp2 isoforms. Journal of cell science, 113(24), 4441-4450.
[1] Zhang, Q., Kang, R., Zeh, III, H. J., Lotze, M. T., & Tang, D. (2013). DAMPs and autophagy: cellular adaptation to injury and unscheduled cell death. Autophagy, 9(4), 451-458.
[1] (a) Deretic, V. (2021). Autophagy in inflammation, infection, and immunometabolism. Immunity, 54(3), 437-453. (b) Tsuji, Y., Kuramochi, M., Golbar, H. M., Izawa, T., Kuwamura, M., & Yamate, J. (2020). Acetaminophen-induced rat hepatotoxicity based on M1/M2-macrophage polarization, in possible relation to damage-associated molecular patterns and autophagy. International Journal of Molecular Sciences, 21(23), 8998. (c) Levy, J. M. M., Towers, C. G., & Thorburn, A. (2017). Targeting autophagy in cancer. Nature Reviews Cancer, 17(9), 528-542.
[1] Levine, B., Mizushima, N., & Virgin, H. W. (2011). Autophagy in immunity and inflammation. Nature, 469(7330), 323-335.
[1] Deretic, V., Saitoh, T., & Akira, S. (2013). Autophagy in infection, inflammation and immunity. Nature Reviews Immunology, 13(10), 722-737.
[1] Moore, M. N. (2020). Lysosomes, autophagy, and hormesis in cell physiology, pathology, and age-related disease. Dose-Response, 18(3), 1559325820934227.
[1] García-Prat, L., Martínez-Vicente, M., Perdiguero, E., Ortet, L., Rodríguez-Ubreva, J., Rebollo, E., … & Muñoz-Cánoves, P. (2016). Autophagy maintains stemness by preventing senescence. Nature, 529(7584), 37-42.
[1] Hara, T., Nakamura, K., Matsui, M., Yamamoto, A., Nakahara, Y., Suzuki-Migishima, R., … & Mizushima, N. (2006). Suppression of basal autophagy in neural cells causes neurodegenerative disease in mice. Nature, 441(7095), 885-889.
[1] Zapata-Muñoz, J., Villarejo-Zori, B., Largo-Barrientos, P., & Boya, P. (2021). Towards a better understanding of the neuro-developmental role of autophagy in sickness and in health. Cell Stress, 5(7), 99.
[1] Nixon, R. A. (2013). The role of autophagy in neurodegenerative disease. Nature medicine, 19(8), 983-997.
[1] Tomoda, T., Yang, K., & Sawa, A. (2020). Neuronal autophagy in synaptic functions and psychiatric disorders. Biological psychiatry, 87(9), 787-796.
[1] Sil, P., Wong, S. W., & Martinez, J. (2018). More than skin deep: autophagy is vital for skin barrier function. Frontiers in immunology, 9, 1376.
[1] Jeong, D., Qomaladewi, N. P., Lee, J., Park, S. H., & Cho, J. Y. (2020). The role of autophagy in skin fibroblasts, keratinocytes, melanocytes, and epidermal stem cells. Journal of Investigative Dermatology, 140(9), 1691-1697.
[1] Richmond, J. M., & Harris, J. E. (2014). Immunology and skin in health and disease. Cold Spring Harbor perspectives in medicine, 4(12), a015339.
[1] Pivarcsi, A., Nagy, I., & Kemeny, L. (2005). Innate immunity in the skin: how keratinocytes fight against pathogens. Current immunology reviews, 1(1), 29-42.
[1] Davidson, S., Coles, M., Thomas, T., Kollias, G., Ludewig, B., Turley, S., … & Buckley, C. D. (2021). Fibroblasts as immune regulators in infection, inflammation and cancer. Nature Reviews Immunology, 21(11), 704-717.
[1] Zhang, H., Xia, M., Li, H., Zeng, X., Jia, H., Zhang, W., & Zhou, J. (2025). Implication of Immunobiological Function of Melanocytes in Dermatology. Clinical Reviews in Allergy & Immunology, 68(1), 30.
[1] Jeong, D., Qomaladewi, N. P., Lee, J., Park, S. H., & Cho, J. Y. (2020). The role of autophagy in skin fibroblasts, keratinocytes, melanocytes, and epidermal stem cells. Journal of Investigative Dermatology, 140(9), 1691-1697.
[1] Ren, H., Zhao, F., Zhang, Q., Huang, X., & Wang, Z. (2022). Autophagy and skin wound healing. Burns & trauma, 10, tkac003.
[1] Li, X., Wu, J., Sun, X., Wu, Q., Li, Y., Li, K., … & Chen, H. (2020). Autophagy reprograms alveolar progenitor cell metabolism in response to lung injury. Stem Cell Reports, 14(3), 420-432.
[1] de la Vega, M. R., Dodson, M., Gross, C., Mansour, H. M., Lantz, R. C., Chapman, E., … & Zhang, D. D. (2016). Role of Nrf2 and autophagy in acute lung injury. Current pharmacology reports, 2, 91-101.
[1] Yin, X., Xin, H., Mao, S., Wu, G., & Guo, L. (2019). The role of autophagy in sepsis: protection and injury to organs. Frontiers in physiology, 10, 1071.
[1] Barnes, P. J., Baker, J., & Donnelly, L. E. (2022). Autophagy in asthma and chronic obstructive pulmonary disease. Clinical Science, 136(10), 733-746.
[1] Mizumura, K., Maruoka, S., Shimizu, T., & Gon, Y. (2018). Autophagy, selective autophagy, and necroptosis in COPD. International journal of chronic obstructive pulmonary disease, 3165-3172.
[1] Araya, J., Kojima, J., Takasaka, N., Ito, S., Fujii, S., Hara, H., … & Kuwano, K. (2013). Insufficient autophagy in idiopathic pulmonary fibrosis. American Journal of Physiology-Lung Cellular and Molecular Physiology, 304(1), L56-L69.
[1] Patel, A. S., Lin, L., Geyer, A., Haspel, J. A., An, C. H., Cao, J., … & Morse, D. (2012). Autophagy in idiopathic pulmonary fibrosis. PloS one, 7(7), e41394.
[1] McAlinden, K. D., Deshpande, D. A., Ghavami, S., Xenaki, D., Sohal, S. S., Oliver, B. G., … & Sharma, P. (2019). Autophagy activation in asthma airways remodeling. American journal of respiratory cell and molecular biology, 60(5), 541-553.
[1] Liu, C., Liu, Y., Chen, H., Yang, X., Lu, C., Wang, L., & Lu, J. (2023). Myocardial injury: where inflammation and autophagy meet. Burns & Trauma, 11, tkac062.
[1] Alcalai, R., Arad, M., Wakimoto, H., Yadin, D., Gorham, J., Wang, L., … & Seidman, C. E. (2021). LAMP2 cardiomyopathy: consequences of impaired autophagy in the heart. Journal of the American Heart Association, 10(17), e018829.
[1] Liu, Y., Wang, Y., Bi, Y., Zhao, Z., Wang, S., Lin, S., … & Mao, J. (2023). Emerging role of mitophagy in heart failure: from molecular mechanism to targeted therapy. Cell Cycle, 22(8), 906-918.
[1] Shen, Y., Liu, X., Shi, J., & Wu, X. (2019). Involvement of Nrf2 in myocardial ischemia and reperfusion injury. International journal of biological macromolecules, 125, 496-502.
[1] Chen, X., Ji, Y., Liu, R., Zhu, X., Wang, K., Yang, X., … & Sun, H. (2023). Mitochondrial dysfunction: roles in skeletal muscle atrophy. Journal of Translational Medicine, 21(1), 503.
[1] Li, P., Ma, Y., Yu, C., Wu, S., Wang, K., Yi, H., & Liang, W. (2021). Autophagy and aging: Roles in skeletal muscle, eye, brain and hepatic tissue. Frontiers in Cell and Developmental Biology, 9, 752962.
[1] Ferraro, E., Giammarioli, A. M., Chiandotto, S., Spoletini, I., & Rosano, G. (2014). Exercise-induced skeletal muscle remodeling and metabolic adaptation: redox signaling and role of autophagy. Antioxidants & redox signaling, 21(1), 154-176.
[1] Weckman, A., Di Ieva, A., Rotondo, F., Syro, L. V., Ortiz, L. D., Kovacs, K., & Cusimano, M. D. (2014). Autophagy in the endocrine glands. J Mol Endocrinol, 52(2), R151-R163.
[1] (a) Afzal, A., Zhang, Y., Afzal, H., Saddozai, U. A. K., Zhang, L., Ji, X. Y., & Khawar, M. B. (2024). Functional role of autophagy in testicular and ovarian steroidogenesis. Frontiers in Cell and Developmental Biology, 12, 1384047. (b) Xu, R., Wang, F., Zhang, Z., Zhang, Y., Tang, Y., Bi, J., … & Tang, Z. (2023). Diabetes‐induced autophagy dysregulation engenders testicular impairment via oxidative stress. Oxidative medicine and cellular longevity, 2023(1), 4365895.
[1] (a) Kong, X., Wang, X., Xia, Q., Hu, Q., Yu, W., Huang, Q., … & Yu, J. (2025). Unveiling the nexus between environmental exposures and testicular damages: revelations from autophagy and oxidative stress imbalance. Cell Death Discovery, 11(1), 258. (b) Jung, H. S., & Lee, M. S. (2010). Role of autophagy in diabetes and mitochondria. Annals of the New York Academy of Sciences, 1201(1), 79-83.
[1] Zhao, X., Jiang, Y., Jiang, T., Han, X., Wang, Y., Chen, L., & Feng, X. (2020). Physiological and pathological regulation of autophagy in pregnancy. Archives of Gynecology and Obstetrics, 302(2), 293-303.
[1] Chen, H., Chen, Y., & Zheng, Q. (2024). The regulated cell death at the maternal-fetal interface: beneficial or detrimental?. Cell Death Discovery, 10(1), 100.
[1] Zhou, P., Wang, J., Wang, J., & Liu, X. (2024). When autophagy meets placenta development and pregnancy complications. Frontiers in Cell and Developmental Biology, 12, 1327167.
[1] Wang, Y., Singh, R., Xiang, Y., & Czaja, M. J. (2010). Macroautophagy and chaperone‐mediated autophagy are required for hepatocyte resistance to oxidant stress. Hepatology, 52(1), 266-277.
[1] Xu, F., Hua, C., Tautenhahn, H. M., Dirsch, O., & Dahmen, U. (2020). The role of autophagy for the regeneration of the aging liver. International journal of molecular sciences, 21(10), 3606.
[1] (a) Cursio, R., Colosetti, P., Codogno, P., Cuervo, A. M., & Shen, H. M. (2015). The role of autophagy in liver diseases: mechanisms and potential therapeutic targets. BioMed Research International, 2015, 480508. (b) Ruart, M., Chavarria, L., Campreciós, G., Suárez-Herrera, N., Montironi, C., Guixé-Muntet, S., … & Hernández-Gea, V. (2019). Impaired endothelial autophagy promotes liver fibrosis by aggravating the oxidative stress response during acute liver injury. Journal of hepatology, 70(3), 458-469.
[1] Jaganjac, M., Milkovic, L., Zarkovic, N., & Zarkovic, K. (2022). Oxidative stress and regeneration. Free Radical Biology and Medicine, 181, 154-165.
[1] Franzini, M., Valdenassi, L., Pandolfi, S., Tirelli, U., Ricevuti, G., & Chirumbolo, S. (2023). The role of ozone as an Nrf2-keap1-ARE activator in the anti-microbial activity and immunity modulation of infected wounds. Antioxidants, 12(11), 1985.
[1] Clavo, B., Rodríguez-Esparragón, F., Rodríguez-Abreu, D., Martínez-Sánchez, G., Llontop, P., Aguiar-Bujanda, D., … & Santana-Rodríguez, N. (2019). Modulation of oxidative stress by ozone therapy in the prevention and treatment of chemotherapy-induced toxicity: review and prospects. Antioxidants, 8(12), 588.
[1] (a) Ristow, M., & Zarse, K. (2010). How increased oxidative stress promotes longevity and metabolic health: The concept of mitochondrial hormesis (mitohormesis). Experimental gerontology, 45(6), 410-418. (b) Calabrese, E. J., Osakabe, N., Di Paola, R., Siracusa, R., Fusco, R., D’Amico, R., … & Calabrese, V. (2023). Hormesis defines the limits of lifespan. Ageing research reviews, 102074.
[1] Hulten, L. M., Holmström, M., & Soussi, B. (1999). Harmful singlet oxygen can be helpful. Free Radical Biology and Medicine, 27(11), 1203-1207.
[1] (a) Bieber, K., Hundt, J. E., Yu, X., Ehlers, M., Petersen, F., Karsten, C. M., … & Ludwig, R. J. (2023). Autoimmune pre-disease. Autoimmunity Reviews, 22(2), 103236. (b) Xiao, Z. X., Miller, J. S., & Zheng, S. G. (2021). An updated advance of autoantibodies in autoimmune diseases. Autoimmunity Reviews, 20(2), 102743.
[1] (a) Tisoncik, J. R., Korth, M. J., Simmons, C. P., Farrar, J., Martin, T. R., & Katze, M. G. (2012). Into the eye of the cytokine storm. Microbiology and molecular biology reviews, 76(1), 16-32. (b) Karki, R., & Kanneganti, T. D. (2021). The ‘cytokine storm’: Molecular mechanisms and therapeutic prospects. Trends in immunology, 42(8), 681-705.
[1] Fridovich, I. (2013). Oxygen: how do we stand it?. Medical Principles and Practice, 22(2), 131-137.
[1] (a) Meyer, J. G., Garcia, T. Y., Schilling, B., Gibson, B. W., & Lamba, D. A. (2019). Proteome and secretome dynamics of human retinal pigment epithelium in response to reactive oxygen species. Scientific reports, 9(1), 15440. (b) Yi, Y., Wang, X. R., Chen, H. T., Huang, W. Y., Feng, L. X., Fang, S. B., & Xiong, G. X. (2023). Development of a serum-free culture method for endothelial cells of the stria vascularis and their pro-inflammatory secretome changes induced by oxidative stress. Clinical and Experimental Otorhinolaryngology, 16(1), 37-48.
[1] Eldh, M., Ekström, K., Valadi, H., Sjöstrand, M., Olsson, B., Jernås, M., & Lötvall, J. (2010). Exosomes communicate protective messages during oxidative stress; possible role of exosomal shuttle RNA. PloS one, 5(12), e15353.
[1] (a) Evren, E., Ringqvist, E., Tripathi, K. P., Sleiers, N., Rives, I. C., Alisjahbana, A., … & Willinger, T. (2021). Distinct developmental pathways from blood monocytes generate human lung macrophage diversity. Immunity, 54(2), 259-275. (b) Tan, S. Y., & Krasnow, M. A. (2016). Developmental origin of lung macrophage diversity. Development, 143(8), 1318-1327.
[1] Chinnery, H. R., McMenamin, P. G., & Dando, S. J. (2017). Macrophage physiology in the eye. Pflügers Archiv-European Journal of Physiology, 469, 501-515.
[1] Puttur, F., Gregory, L. G., & Lloyd, C. M. (2019). Airway macrophages as the guardians of tissue repair in the lung. Immunology and cell biology, 97(3), 246-257.
[1] Rahman, I., & Adcock, I. M. (2006). Oxidative stress and redox regulation of lung inflammation in COPD. European respiratory journal, 28(1), 219-242.
[1] Arora, S., Dev, K., Agarwal, B., Das, P., & Syed, M. A. (2018). Macrophages: Their role, activation and polarization in pulmonary diseases. Immunobiology, 223(4-5), 383-396.
[1] Ogura, M., & Kitamura, M. (1998). Oxidant stress incites spreading of macrophages via extracellular signal-regulated kinases and p38 mitogen-activated protein kinase. The Journal of Immunology, 161(7), 3569-3574.
[1] Reichard, A., Wanner, N., Farha, S., & Asosingh, K. (2023). Hematopoietic stem cells and extramedullary hematopoiesis in the lungs. Cytometry Part A, 103(12), 967-977.
[1] (a) Lämmermann, T., & Sixt, M. (2008). The microanatomy of T‐cell responses. Immunological reviews, 221(1), 26-43. (b) Chinnery, H. R., McMenamin, P. G., & Dando, S. J. (2017). Macrophage physiology in the eye. Pflügers Archiv-European Journal of Physiology, 469, 501-515.
[1] (a) Andersson, D. A., Gentry, C., Moss, S., & Bevan, S. (2008). Transient receptor potential A1 is a sensory receptor for multiple products of oxidative stress. Journal of Neuroscience, 28(10), 2485-2494. (b) Kozai, D., Ogawa, N., & Mori, Y. (2014). Redox regulation of transient receptor potential channels. Antioxidants & redox signaling, 21(6), 971-986.
[1] (a) Tracey, K. J. (2002). The inflammatory reflex. Nature, 420(6917), 853-859. (b) Pavlov, V. A., & Tracey, K. J. (2017). Neural regulation of immunity: molecular mechanisms and clinical translation. Nature neuroscience, 20(2), 156-166.
[1] (a) Antunes, G. L., Silveira, J. S., Kaiber, D. B., Luft, C., da Costa, M. S., Marques, E. P., … & da Cunha, A. A. (2020). Cholinergic anti‐inflammatory pathway confers airway protection against oxidative damage and attenuates inflammation in an allergic asthma model. Journal of Cellular Physiology, 235(2), 1838-1849. (b) Kelly, M. J., Breathnach, C., Tracey, K. J., & Donnelly, S. C. (2022). Manipulation of the inflammatory reflex as a therapeutic strategy. Cell Reports Medicine, 3(7).
[1] Kozai, D., Ogawa, N., & Mori, Y. (2014). Redox regulation of transient receptor potential channels. Antioxidants & redox signaling, 21(6), 971-986.
[1] Gupta, A. K., Gupta, S., Mehan, S., Khan, Z., Das Gupta, G., & Narula, A. S. (2025). Exploring the connection between BDNF/TrkB and AC/cAMP/PKA/CREB signaling pathways: potential for neuroprotection and therapeutic targets for neurological disorders. Molecular Neurobiology, 62(11), 14627-14659.
[1] Bellavance, M. A., & Rivest, S. (2014). The HPA–immune axis and the immunomodulatory actions of glucocorticoids in the brain. Frontiers in immunology, 5, 136.
[1] Sockrider, M., & Fussner, L. (2020). What is asthma?. American journal of respiratory and critical care medicine, 202(9), P25-P26.
[1] Venkatesan, P. (2023). 2023 GINA report for asthma. The Lancet Respiratory Medicine, 11(7), 589.
[1] To be published
[1] Adeloye, D., Song, P., Zhu, Y., Campbell, H., Sheikh, A., & Rudan, I. (2022). Global, regional, and national prevalence of, and risk factors for, chronic obstructive pulmonary disease (COPD) in 2019: a systematic review and modelling analysis. The Lancet Respiratory Medicine, 10(5), 447-458.
[1] Erpenbach, K. H., Brailey, M. A., & Stentiford, N. Bronchial Asthma and Chronic Obstructive Lung Diseases: Improvement of the 6. minute walking test and the lung function by inhaling activated air produced by the SOE. TIE therapeutic inhalation equipment.
[1] Duan, K. I., Birger, M., Au, D. H., Spece, L. J., Feemster, L. C., & Dieleman, J. L. (2023). Health Care Spending on Respiratory Diseases in the United States, 1996–2016. American Journal of Respiratory and Critical Care Medicine, 207(2), 183-192.
[1] Paulson, K. R., Kamath, A. M., Alam, T., Bienhoff, K., Abady, G. G., Abbas, J., … & Chanie, W. F. (2021). Global, regional, and national progress towards Sustainable Development Goal 3.2 for neonatal and child health: all-cause and cause-specific mortality findings from the Global Burden of Disease Study 2019. The Lancet, 398(10303), 870-905.
[1] Gordon, B. R. (2008). Asthma history and presentation. Otolaryngologic Clinics of North America, 41(2), 375-385.
[1] Burgel, P. R. (2012). Chronic cough and sputum production: a clinical COPD phenotype?. European Respiratory Journal, 40(1), 4-6.
[1] Diehr, P., Wood, R. W., Bushyhead, J., Krueger, L., Wolcott, B., & Tompkins, R. K. (1984). Prediction of pneumonia in outpatients with acute cough—a statistical approach. Journal of chronic diseases, 37(3), 215-225.
[1] Tollerud, D. J., O’connor, G. T., Sparrow, D., & Weiss, S. T. (1991). Asthma, hay fever, and phlegm production associated with distinct patterns of allergy skin test reactivity, eosinophilia, and serum IgE levels. The Normative Aging Study. Am Rev Respir Dis, 144(4), 776-81.
[1] Heijdra, Y. F., Pinto-Plata, V. M., Kenney, L. A., Rassulo, J., & Celli, B. R. (2002). Cough and phlegm are important predictors of health status in smokers without COPD. Chest, 121(5), 1427-1433.
[1] (a) Pitts, T., Bolser, D., Rosenbek, J., Troche, M., & Sapienza, C. (2008). Voluntary cough production and swallow dysfunction in Parkinson’s disease. Dysphagia, 23, 297-301. (b) Heijdra, Y. F., Pinto-Plata, V. M., Kenney, L. A., Rassulo, J., & Celli, B. R. (2002). Cough and phlegm are important predictors of health status in smokers without COPD. Chest, 121(5), 1427-1433. (c) Davies, D. (1974). Disability and coal workers’ pneumoconiosis. British medical journal, 2(5920), 652. (d) Warren, C. P. (1979). Acute respiratory failure and tracheal obstruction in the elderly with benign goitres. Canadian Medical Association Journal, 121(2), 191.
[1] Whitsett, J. A., & Alenghat, T. (2015). Respiratory epithelial cells orchestrate pulmonary innate immunity. Nature immunology, 16(1), 27-35.
[1] Voynow, J. A., & Rubin, B. K. (2009). Mucins, mucus, and sputum. Chest, 135(2), 505-512.
[1] (a) Biswas, S. K., & Rahman, I. (2009). Environmental toxicity, redox signaling and lung inflammation: the role of glutathione. Molecular aspects of medicine, 30(1-2), 60-76. (b) Shao, M. X., & Nadel, J. A. (2005). Dual oxidase 1-dependent MUC5AC mucin expression in cultured human airway epithelial cells. Proceedings of the National Academy of Sciences, 102(3), 767-772.
[1] Price, M. E., & Sisson, J. H. (2019). Redox regulation of motile cilia in airway disease. Redox Biology, 27, 101146.
[1] Rancourt, R. C., Lee, R. L., O’Neill, H., Accurso, F. J., & White, C. W. (2007). Reduced thioredoxin increases proinflammatory cytokines and neutrophil influx in rat airways: modulation by airway mucus. Free Radical Biology and Medicine, 42(9), 1441-1453.
[1] Zhang, M., Wang, J., Liu, R., Wang, Q., Qin, S., Chen, Y., & Li, W. (2024). The role of Keap1-Nrf2 signaling pathway in the treatment of respiratory diseases and the research progress on targeted drugs. Heliyon.
[1] (a) Barnes, P. J. (2008). The cytokine network in asthma and chronic obstructive pulmonary disease. The Journal of clinical investigation, 118(11), 3546-3556. (b) Xu, W., Zhao, T., & Xiao, H. (2020). The implication of oxidative stress and AMPK-Nrf2 antioxidative signaling in pneumonia pathogenesis. Frontiers in Endocrinology, 11, 400.
[1] Kume, H., Yamada, R., Sato, Y., & Togawa, R. (2023). Airway smooth muscle regulated by oxidative stress in COPD. Antioxidants, 12(1), 142.
[1] Michaeloudes, C., Abubakar-Waziri, H., Lakhdar, R., Raby, K., Dixey, P., Adcock, I. M., … & Chung, K. F. (2022). Molecular mechanisms of oxidative stress in asthma. Molecular aspects of medicine, 85, 101026.
[1] (a) Zhou, F., Yu, T., Du, R., Fan, G., Liu, Y., Liu, Z., … & Cao, B. (2020). Clinical course and risk factors for mortality of adult inpatients with COVID-19 in Wuhan, China: a retrospective cohort study. The lancet, 395(10229), 1054-1062. (b) George, P. M., Barratt, S. L., Condliffe, R., Desai, S. R., Devaraj, A., Forrest, I., … & Spencer, L. G. (2020). Respiratory follow-up of patients with COVID-19 pneumonia. Thorax, 75(11), 1009-1016.
[1] Wu, Z., & McGoogan, J. M. (2020). Characteristics of and important lessons from the coronavirus disease 2019 (COVID-19) outbreak in China: summary of a report of 72 314 cases from the Chinese Center for Disease Control and Prevention. jama, 323(13), 1239-1242.
[1] “Post COVID-19 condition (long COVID).” World Health Organization (WHO), 26 February 2025, https://www.who.int/news-room/fact-sheets/detail/post-covid-19-condition-(long-covid). Accessed 2 August 2025.
[1] (a) Nalbandian, A., Sehgal, K., Gupta, A., Madhavan, M. V., McGroder, C., Stevens, J. S., … & Wan, E. Y. (2021). Post-acute COVID-19 syndrome. Nature medicine, 27(4), 601-615. (b) Yong, S. J. (2021). Persistent brainstem dysfunction in long-COVID: a hypothesis. ACS chemical neuroscience, 12(4), 573-580.
[1] Sakaguchi, R., & Mori, Y. (2020). Transient receptor potential (TRP) channels: Biosensors for redox environmental stimuli and cellular status. Free Radical Biology and Medicine, 146, 36-44.
[1] Zielinski, M. R., McKenna, J. T., & McCarley, R. W. (2016). Functions and mechanisms of sleep. AIMS neuroscience, 3(1), 67.
[1] Peliciari-Garcia, R. A., Darley-Usmar, V., & Young, M. E. (2018). An overview of the emerging interface between cardiac metabolism, redox biology and the circadian clock. Free radical biology and medicine, 119, 75-84.
[1] (a) Reimund, E. (1994). The free radical flux theory of sleep. Medical hypotheses, 43(4), 231-233. (b) Villafuerte, G., Miguel-Puga, A., Murillo Rodríguez, E., Machado, S., Manjarrez, E., & Arias-Carrión, O. (2015). Sleep deprivation and oxidative stress in animal models: a systematic review. Oxidative medicine and cellular longevity, 2015(1), 234952.
[1] (a) Bryndin, E., & Bryndina, I. (2020). Self healing of healthy condition at cellular level. Medical Case Reports and Reviews, 3, 1-4. (b) Adam, K., & Oswald, I. (1984). Sleep helps healing. British medical journal (Clinical research ed.), 289(6456), 1400.
[1] (a) Garbarino, S., Lanteri, P., Bragazzi, N. L., Magnavita, N., & Scoditti, E. (2021). Role of sleep deprivation in immune-related disease risk and outcomes. Communications biology, 4(1), 1304. (b) Tobaldini, E., Costantino, G., Solbiati, M., Cogliati, C., Kara, T., Nobili, L., & Montano, N. (2017). Sleep, sleep deprivation, autonomic nervous system and cardiovascular diseases. Neuroscience & Biobehavioral Reviews, 74, 321-329.
[1] Wilking, M., Ndiaye, M., Mukhtar, H., & Ahmad, N. (2013). Circadian rhythm connections to oxidative stress: implications for human health. Antioxidants & redox signaling, 19(2), 192-208.
[1] Atrooz, F., & Salim, S. (2020). Sleep deprivation, oxidative stress and inflammation. Advances in protein chemistry and structural biology, 119, 309-336.
[1] Patki, G., Solanki, N., Atrooz, F., Allam, F., & Salim, S. (2013). Depression, anxiety-like behavior and memory impairment are associated with increased oxidative stress and inflammation in a rat model of social stress. Brain research, 1539, 73-86.
[1] Mattson, M. P. (2008). Hormesis defined. Ageing research reviews, 7(1), 1-7.
[1] Besedovsky, L., Lange, T., & Haack, M. (2019). The sleep-immune crosstalk in health and disease. Physiological reviews.
[1] (a) Deary, V., Ellis, J. G., Wilson, J. A., Coulter, C., & Barclay, N. L. (2014). Simple snoring: not quite so simple after all?. Sleep medicine reviews, 18(6), 453-462. (b) Sarkis, L. M., Jones, A. C., Ng, A., Pantin, C., Appleton, S. L., & MacKay, S. G. (2023). Australasian Sleep Association position statement on consensus and evidence based treatment for primary snoring. Respirology, 28(2), 110-119.
[1] Gonzalez-Rothi, E. J., Lee, K. Z., Dale, E. A., Reier, P. J., Mitchell, G. S., & Fuller, D. D. (2015). Intermittent hypoxia and neurorehabilitation. Journal of applied physiology, 119(12), 1455-1465.
[1] Swart, M. L., Van Schagen, A. M., Lancee, J., & Van Den Bout, J. (2013). Prevalence of nightmare disorder in psychiatric outpatients. Psychotherapy and psychosomatics, 82(4), 267-268.
[1] (a) Levin, R., & Nielsen, T. A. (2007). Disturbed dreaming, posttraumatic stress disorder, and affect distress: a review and neurocognitive model. Psychological bulletin, 133(3), 482. (b) Gieselmann, A., Ait Aoudia, M., Carr, M., Germain, A., Gorzka, R., Holzinger, B., … & Pietrowsky, R. (2019). Aetiology and treatment of nightmare disorder: State of the art and future perspectives. Journal of sleep research, 28(4), e12820.
[1] Krakow, B., & Zadra, A. (2006). Clinical management of chronic nightmares: imagery rehearsal therapy. Behavioral Sleep Medicine, 4(1), 45–70.
[1] Germain, A., Buysse, D. J., & Nofzinger, E. (2008). Sleep-specific mechanisms underlying posttraumatic stress disorder: integrative review and neurobiological hypotheses. Sleep medicine reviews, 12(3), 185-195.
[1] Hsieh, C. F., Huang, C. T., Chang, C. C., & Hung, T. P. (2025). Singlet Oxygen Energy for Enhancing Physiological Function and Athletic Performance. Bioengineering, 12(2), 118.
[1] Martinez-Canton, M., Galvan-Alvarez, V., Martin-Rincon, M., Calbet, J. A., & Gallego-Selles, A. (2024). Unlocking peak performance: The role of Nrf2 in enhancing exercise outcomes and training adaptation in humans. Free Radical Biology and Medicine, 224, 168-181.
[1] Taskin, S., Celik, T., Demiryurek, S., Turedi, S., & Taskin, A. (2022). Effects of different-intensity exercise and creatine supplementation on mitochondrial biogenesis and redox status in mice. Iranian Journal of Basic Medical Sciences, 25(8), 1009.
[1] Sun, Y., Jin, L., Qin, Y., Ouyang, Z., Zhong, J., & Zeng, Y. (2024). Harnessing mitochondrial stress for health and disease: Opportunities and challenges. Biology, 13(6), 394.
[1] Viollet, B. (2017). The energy sensor AMPK: adaptations to exercise, nutritional and hormonal signals. Hormones, metabolism and the benefits of exercise, 13.
[1] Meng, Q., & Su, C. H. (2024). The impact of physical exercise on oxidative and nitrosative stress: balancing the benefits and risks. Antioxidants, 13(5), 573.
[1] World Health Organization. (2023). Accelerating anaemia reduction: Acomprehensive framework for action. World Health Organization.
[1] Weiss, G., Ganz, T., & Goodnough, L. T. (2019). Anemia of inflammation. Blood, The Journal of the American Society of Hematology, 133(1), 40-50.
[1] Shaw, P., & Chattopadhyay, A. (2020). Nrf2–ARE signaling in cellular protection: Mechanism of action and the regulatory mechanisms. Journal of Cellular Physiology, 235(4), 3119-3130.
[1] Dewhirst, M. W. (2009). Relationships between cycling hypoxia, HIF-1, angiogenesis and oxidative stress. Radiation research, 172(6), 653-665.
[1] Wang, J., & Pantopoulos, K. (2011). Regulation of cellular iron metabolism. Biochemical Journal, 434(3), 365-381.
[1] ORINO, K., LEHMAN, L., TSUJI, Y., AYAKI, H., TORTI, S. V., & TORTI, F. M. (2001). Ferritin and the response to oxidative stress. Biochemical Journal, 357(1), 241-247.
[1] Wilson, B. E., Jacob, S., Yap, M. L., Ferlay, J., Bray, F., & Barton, M. B. (2019). Estimates of global chemotherapy demands and corresponding physician workforce requirements for 2018 and 2040: a population-based study. The Lancet Oncology, 20(6), 769-780.
[1] Sano, M., & Fukuda, K. (2008). Activation of mitochondrial biogenesis by hormesis. Circulation research, 103(11), 1191-1193.
[1] Clavo, B., Rodríguez-Esparragón, F., Rodríguez-Abreu, D., Martínez-Sánchez, G., Llontop, P., Aguiar-Bujanda, D., … & Santana-Rodríguez, N. (2019). Modulation of oxidative stress by ozone therapy in the prevention and treatment of chemotherapy-induced toxicity: review and prospects. Antioxidants, 8(12), 588.
[1] Li, N., & Karin, M. (1999). Is NF‐κB the sensor of oxidative stress?. The FASEB Journal, 13(10), 1137-1143.
[1] (a) Pradat, P. F., & Delanian, S. (2013). Late radiation injury to peripheral nerves. Handbook of clinical neurology, 115, 743-758. (b) Pariset, E., Malkani, S., Cekanaviciute, E., & Costes, S. V. (2021). Ionizing radiation-induced risks to the central nervous system and countermeasures in cellular and rodent models. International Journal of Radiation Biology, 97(sup1), S132-S150. (c) Azzam, P., Mroueh, M., Francis, M., Abou Daher, A., & Zeidan, Y. H. (2020). Radiation-induced neuropathies in head and neck cancer: prevention and treatment modalities. ecancermedicalscience, 14, 1133.
[1] Yoon, K. C., Jeong, I. Y., Park, Y. G., & Yang, S. Y. (2007). Interleukin-6 and tumor necrosis factor-α levels in tears of patients with dry eye syndrome. Cornea, 26(4), 431-437.
[1] Pflugfelder, S. C., & de Paiva, C. S. (2017). The pathophysiology of dry eye disease: what we know and future directions for research. Ophthalmology, 124(11), S4-S13.
[1] Böhm, E. W., Buonfiglio, F., Voigt, A. M., Bachmann, P., Safi, T., Pfeiffer, N., & Gericke, A. (2023). Oxidative stress in the eye and its role in the pathophysiology of ocular diseases. Redox biology, 68, 102967.
[1] Grimwood, S. J. (2024). Profiling the Physiological and Psychological Effectiveness of Singlet Oxygen in the Management of Chronic Obstructive Pulmonary Disease (COPD) (Doctoral dissertation, University of Derby (United Kingdom)).
[1] (a) Price, D. D., Finniss, D. G., & Benedetti, F. (2008). A comprehensive review of the placebo effect: recent advances and current thought. Annu. Rev. Psychol., 59(1), 565-590. (b) Enck, P., Benedetti, F., & Schedlowski, M. (2008). New insights into the placebo and nocebo responses. Neuron, 59(2), 195-206.
[1] (a) Colloca, L., & Barsky, A. J. (2020). Placebo and nocebo effects. New England Journal of Medicine, 382(6), 554-561. (b) Benedetti, F., & Amanzio, M. (2013). Mechanisms of the placebo response. Pulmonary pharmacology & therapeutics, 26(5), 520-523.
[1] Liu, S., Pi, J., & Zhang, Q. (2022). Signal amplification in the KEAP1-NRF2-ARE antioxidant response pathway. Redox biology, 54, 102389.